In-Vitro Phenotypic Response of Human Osteoblasts to Different Titanium Surface Roughness
Muataz A Osman1,2,3,5*, Rasha A Alamoush4, Evgeny Kushnerev1,5, Kevin G Seymour1, Susan Shawcross5 and Julian M Yates1
1Division of Dentistry, School of Medical Sciences, Coupland 3 Building, University of Manchester, Oxford Road, Manchester, M13 9PL, United Kingdom
2Periodontology Department, Faculty of Dentistry, The University of Benghazi, Benghazi-Libya
3Restorative Department, Faculty of Dentistry, Libyan International Medical University, Benghazi-Libya
4Prosthodontic Department, School of Dentistry, University of Jordan, Amman, Jorda
5Blond McIndoe Laboratories, Division of Cell Matrix Biology & Regenerative Medicine, Faculty of Biology, Medicine & Health, The University of Manchester, 3.106 Stopford Building, Oxford Road, Manchester M13 9PT, United Kingdom
Submission: June 05, 2022; Published: June 13, 2022
*Corresponding author: Muataz A Osman, Division of Dentistry, School of Medical Sciences, Coupland 3 Building, University of Manchester, Oxford Road, Manchester, M13 9PL, United Kingdom
How to cite this article: Muataz A Osman, Rasha A Alamoush, Evgeny Kushnerev, Kevin G Seymour, Susan Shawcross, Julian M Yates. In-Vitro Phenotypic Response of Human Osteoblasts to Different Titanium Surface Roughness. Adv Dent & Oral Health. 2022; 15(4): 555916. DOI: 10.19080/ADOH.2022.15.555916
Abstract
Objectives: This study aimed to investigate human osteoblasts (HOB) responses towards different titanium (Ti) implant surface roughness.
Methods: Four Ti surface roughness degrees were investigated on a micrometer roughness scale: smooth surface (S: 0.08-0.1μm), minimally rough surface (MM: 0.3-0.5μm), moderately rough surface (MR: 1.2-1.4μm), and rough surface (R: 3.3-3.7μm). HOB cells were cultured, expanded, and maintained according to the supplier’s protocol. Cell proliferation and cytotoxicity were evaluated at day 1, 3, 5, and 10 using Alamar Blue and lactate dehydrogenase colorimetric assays. Data were analysed via two-way ANOVA, one-way ANOVA and Tukey’s post hoc test (p = 0.05 for all tests).
Results: There was no significant difference in cell proliferation or cytotoxicity of the HOB cells in contact with the different Ti surface roughness degrees. There was however a significant time effect on cell proliferation (p < 0.0001) with different exposure durations, for each roughness degree. Furthermore, a non-significant positive correlation between proliferation and cytotoxicity in all investigated surface roughness degrees was observed.
Significance: All investigated roughness degrees showed comparable HOB proliferation with MR surface presenting the highest percentage followed by R, MM, and S, respectively. S surface showed the highest cytotoxic effect on HOB; however, it did not reach the cytotoxic level as suggested by ISO for any medical device to be considered cytotoxic.
Keywords:Surface roughness; Titanium; Titanium surface roughness; Human osteoblasts; Cell Proliferation; Cytotoxicity
Abbreviations:HOB: Investigate Human Osteoblasts; VSI: Vertical Scanning Interferometry; OGM: Osteoblast Growth Medium; Sa: Surface roughness; Sv: Maximum Valley Depth from Mean Plane; Ssk: Skewness; Sdr: Developed Surface Area; Sds: Density of Summits; SD: Standard Deviation; HA: Hydroxyapatite
1. Introduction
Since the time of the pioneering research of Branemark, Albrektsson, Zarb, and others in the field of osseointegration in the 1950s -1980s [1-4], the material of choice when making dental and Orthopaedic implants is still Ti and its alloys, even though increasing attention towards alternative materials has grown in recent years, including zirconia (Zr), polyether ether ketone (PEEK) and others [5]. Ti implants are still considered the gold standard for oral implantology, primarily due to their excellent biocompatibility, mechanical properties, the ability to form an intimate bone-implant contact with living bone by a cement-free connection at the light-microscopic level, as well as its corrosion resistance [5-8]. These characteristics ensure the successful long-term function of the anchored prosthetic restoration [9]. An instantaneously forming passive Ti oxide (TiO₂) layer on the surface leads to corrosion resistance and enhanced biocompatibility [10-12]. Furthermore, Ti is amenable to alterations and changes in its physical and chemical properties, by changing the surface oxide composition, thickness and microtopography. These together, make Ti a suitable material for enhancement via surface modification [13].
Biocompatibility is a material’s feature that prevents the development of an immune response, foreign body reaction, and/or rejection when introduced to the human body [14]. The primary interaction between any material and the host tissues starts with a thin zone of rapid protein adsorption and the formation of a connective tissue interface [13]. This interaction is influenced by the physical and chemical properties of the implant such as surface roughness, structure, composition, defects, as well as the thickness of the TiO₂ layer. These properties are also critical for the long-term success and survival rate of any dental implant [13,15]. Biological materials can be classified into: (1) bio-tolerant, where a thin fibrous tissue layer is formed; (2) bio-inert, such as Ti, which can undergo direct bone contact under osteo-permissive conditions, and finally (3) bio-active materials, such as calcium phosphate ceramics, which can have high degree of direct contact with the surrounding bone due to the release of free calcium and phosphate ions at the implant-bone interface [16]. More recently, these have been re-categorized as 1st generation (structural, biocompatible), including, Ti, 2nd generation (bioactive), including, hydroxyapatite (HA) and bioactive glass, and 3rd generation (reproducible molecular control), including, nano-topography [16,17]. The biocompatibility and inertness of Ti and its alloys are related to the presence of the TiO₂ layer that interacts favorably with water ions and serum proteins, as well as their enhanced corrosion resistance property [18,19].
Several in-vitro studies have illustrated that successful osseointegration between bone and Ti dental implants depends on the implant macro- and micro-surface topography, chemical composition as well as surface energy [20,21]. Surface roughness is an important factor that can influence the biological interactions between cells, tissues, and biomaterials [21]. Suggestions have been made that dental implant and abutment surface topography and roughness degrees may have an impact on peri-implant soft tissue health and bone levels, and consequently may affect the incidence of biological complications such as inflammation, infection, bone loss, implant mobility and eventually implant loss [22]. Various physical, chemical, and mechanical methods have been applied to Ti-based biomaterials to produce micrometer surface structures to help stimulate bone-implant contact. The most common surface treatments include machining, milling, acidetching, grit-blasting, electrochemical methods, and deposition of different ions, proteins, and antimicrobial agents [20]. In-vivo studies have demonstrated that bone tends to form preferentially on R surfaces, whereas connective tissue forms more favorably on S surfaces [23,24]. Considering the trans-gingival nature of dental implants, forming a number of simultaneous interfaces with the host biological system, it is important to understand the different cells and tissues involved in this process and how these interfaces may affect their interaction with certain tissues. These interfaces consist of: (1) the implant body-bone interface, (2) the soft tissue interface at the level of implant neck/platform, and finally (3) the soft tissue interface at the junction of the supra-gingival region [5]. Each surface of the dental implant should be optimized to fulfil the different demands of the respective interfaces. For example, at the implant body level, osteogenic properties are required to optimize bone contact, formation, maturation, and subsequent osseointegration, whilst at the soft tissue interface,gingival attachment with cell-adhesion abilities for fibroblasts and keratinocytes is essential to ensure a tight epithelial seal around the implant neck and its abutment in order to prevent bacterial infiltration and inflammation [25].
After implant placement, several crucial cellular interactions create a strong bone-to-implant connection. It is essential that cells adhere to the implant surface and the surface roughness of dental implants can have a significant influence on HOB adhesion in the early phase of healing, as well as improving and accelerating the osseointegration process [22]. Other important factors are biocompatibility and resistance to bacterial infiltration and contamination [21]. The bio-inert property of Ti is ensured by the protective layer of TiO₂ that forms on its surface. This layer prevents the penetration of metal compounds, and calcium and phosphate ions are readily adherent to the surface, which is necessary for the formation and maturation of the mineralized bone structure. Since the presence of this layer alone is not sufficient for the biocompatibility of Ti, a suitable surface finish and roughness is required to help create and enhance a strong bone-to-implant connection and improve the long-term success following implant therapy [21,26].
Surface roughness are values that can either be calculated on a profile (line/2D) or on a surface (area/3D). The profile roughness parameters are Ra, Rq, etc. The area roughness parameters are Sa, Sq, which give more significant values (that define the height of the surface topography) the reason why they have been employed recently as the preferable parameters to describe surface roughness in implant dentistry [27]. Dental implant surfaces can be classified into four different groups according to their surface roughness. S surfaces with a Sa value of less than 0.3μm, MM with a Sa values of 0.3 to less than 1.0μm, MR where Sa values are between 1.0–2.0μm, and finally, R surfaces where the Sa value is more than 2.0μm [28]. Despite the reported clinical benefits and advantages of the currently available, surface-modified implants, some scientific reports suggest that surface roughness may play a key role in the accelerated development of bone-implant connection in the short term, and also of peri-implant soft and hard tissue damage which could be either reversible (periimplant mucositis) or irreversible (peri-implantitis), in the long term [22,29]. This study is aims to investigate human osteoblast (HOB) response towards various Ti (Ti6Al-4V) implant surface roughness (created by simple industrial milling machine), and to determine if surface roughness (created by simple industrial milling machine) influences early-stage of HOB proliferation in the same way other surface treatments do, which in turn may potentially influence bony healing.
The null hypotheses are that:
a) There is no difference in the proliferation of the HOB cells in contact with Ti discs of various degrees of surface roughness,
b) There is no difference in the cytotoxicity of the HOB cells in contact with Ti discs of various degrees of surface roughness and that.
c) There is no effect on either proliferation or cytotoxicity of the HOB cells with different exposure times for each surface roughness degree.
2. Materials and Methods
2.1. Specimens Preparation

Eighty-eight Ti Ti6Al-4V (grade 4 cert:20) discs (Figure 1) were produced and received from a commercial supplier (GC Tech. Europe GmbH, Harkortstr.2, D-58339 Breckerfeld, Germany), with four different roughness groups: S (n =22), MM (n =22), MR (n = 22), and R (n =22) R. The discs’ dimensions were: 12mm in diameter and 1mm thickness modified on both sides. Different surface roughness degrees were produced using a milling machine utilising a milling bur that can be adjusted between 0.002 to 0.05 mm. Discs were then washed using industrial ultrasound equipment (ATU Ultrasonidos®, Brussels, Belgium) in a soapy solution at 60°C to eliminate traces of oil and/or other residue derived from the machining process without damaging the surfaces. Discs were then dried in a forced convection oven (JP Selecta®, Brussels, Belgium).
2.2. Surface Roughness Measurements
The surface roughness of the Ti discs was measured using a 3D, non-contact, high resolution contour GT-K optical surface profiler (Veeco Contour GT™, Tuscon AZ, USA). To perform the measurement, a vertical scanning interferometry (VSI) mode was selected. The 50x objective lens that provides an area of 174.7 (x) and 132 (y) microns was used. A Gaussian regression filter with a short wavelength cut off of 25μm (0.025mm) was applied before determining the surface roughness parameters. Two samples from each group investigated surface were measured at three random points on each side (six points in total per disc). Five different surface roughness parameters were measured as per the data on the profilometer software: mean surface roughness (Sa), maximum valley depth from mean plane (Sv), skewness (Ssk), developed surface area (Sdr %), and density of summits (Sds). However, the Sa value “the arithmetic means of values above and below a mean plane” was used to represent the Ti surface roughness as recommended by dental implant research methodology and scientific papers [30,31].
2.3. Surface Morphology Analysis
The surface of (n =2) discs of each group were also examined using a scanning electron microscope (SEM) (Quanta™ FEG 250 SEM, Edificio I+D - Campus Río Ebro C/ Mariano Esquillor s/n 50018 Zaragoza – Spain). The images were obtained using the following parameters: 500μm magnification, accelerating voltage of 20kV, spot size of 3.0, and working distance (WD) of 7.6-7.9mm. The use of the SEM in this study allowed the imaging of the surface roughness degree of the Ti discs and visually observe the difference between the different investigated groups.
2.4. HOB Cell Culture Preparation
Primary human osteoblasts (HOB) (Lot No.: 445Z009.2) were obtained from Promo Cell (Heidelberg, Germany), which were derived from human hipbone (femur of a 61-year-old fit and healthy female) biopsies and were cultured according to Promo Cell guidelines and the procedure described by Alamoush et al. [32]. Cells were cultured in osteoblast growth medium (OGM) and mixed with Supplement Mix supplied by the same company. Culture medium was free of antibiotics as per the manufacturer’s recommendations. The cells were expanded and passaged at regular periods based on their growth characteristics and manufacturer’s protocol. Incubation was performed at 37°C, and 5% CO₂ (Panasonic CO₂ Incubator, MCO-170AIC, Panasonic Healthcare Co. Ltd, Japan). Ti discs were placed in 24-well plates in 500μL OGM and incubated for 24 hours before seeding the cells. Once confluent, cells were detached using 0.25% Trypsin- EDTA (Gibco™, Life Technologies, Inc. Canada). Cells were then counted using a Millipore Scepter counter (Merck Millipore, UK) and 5 × 10⁴ cells seeded on each disc in a 24-well culture plate (Corning Costar Ultra-Low attachment multi-well plates (Corning Inc., Corning, NY) in 500μL of complete growth medium. All experiments were performed using appropriate controls with biological and instrumental triplicates and replicated at least 3 times.
2.5. Cell Viability
Cellular viability of 100% was attributed to control wells, where cells were cultured with no Ti discs (low control (LC) or positive growth control). Cellular viability was quantified via a colorimetric assay using invitrogen alamarBlue™ Cell Viability Reagent, DAL1100, Lot: 2120063 (Life Technologies Corporation, Thermo Fisher Scientific, IL, USA). Cell viability was measured at four-time points, day 1, 3, 5 and 10 of cell growth. HOB at each time point were exposed to alamarBlue™ (1:10, reagent: OGM) for 1 hour at 37°C. Then 100μL of supernatant was transferred into a 96-well plate in triplicates for analysis at each time point. The 96- well plate (Corning Costar Ultra-Low attachment multi-well plates Corning Inc., Corning, NY) was read with a UVM 340-microplate reader at 570nm and 600nm (ASYS, Scientific laboratory supplies). Cell viability was calculated according to the following equation [33]:
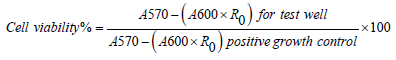
Where A570 and A600 are absorbance at 570 and 600nm respectively, and R is the correction factor calculated from (A570/ A600) of the positive growth control.
2.6. Cytotoxicity
Cytotoxicity of the tested materials was investigated using a CyQUANT™ LDH Cytotoxicity Assay kit, C20301 (Thermo Fisher Scientific, IL, USA). Cytotoxicity on HOB was measured at 4 time points, day 1, 3, 5, and 10. At each of the four time points, as per the company’s protocol, 50μL of lysis buffer (contain membranolytic particles) were added to the specific time point wells (maximum LDH release, high control, HC), and 50μL of sterile-filtered, BioReagent water (SIGMA-ALDRICHᴿ, Life Science, UK) added to the low-control wells (spontaneous LDH release) and the plates incubated at 37°C in 5% CO₂ for 45 minutes. The cytotoxicity was then measured using 50μL of the supernatant and 50μL of LDH cell reaction solution incubated for 30 min at room temperature in the dark. The reaction was stopped using 50μL of the LDH kit stop solution. The 96-well plate (Corning Costar Ultra-Low attachment multi-well plates Corning Inc., Corning, NY) was read with a UVM 340-microplate reader at 490nm subtracted from 680nm (ASYS, Scientific laboratory supplies) and cytotoxicity was calculated according to the following equation [34]:

Where specimen treated LDH activity is the LDH amount expressed by cells cultured with Ti discs, maximum LDH activity is the LDH amount expressed by cells treated with lysis buffer, and the spontaneous LDH activity is the LDH amount expressed by cells treated with sterile water
2.7. Statistical Analysis
Data were analysed using statistical software (GraphPad Prism version 9.1.2 (226)) and found to be normally distributed (Shapiro-Wilk’s test). Two-way ANOVA was performed for roughness effect, time effect and their interaction, followed by one-way ANOVA and Tukey’s multiple comparisons performed to compare cell viability and cytotoxicity for different roughness at each time point (Significant p value = 0.05 for all tests).
3. Results
3.1. Surface Roughness
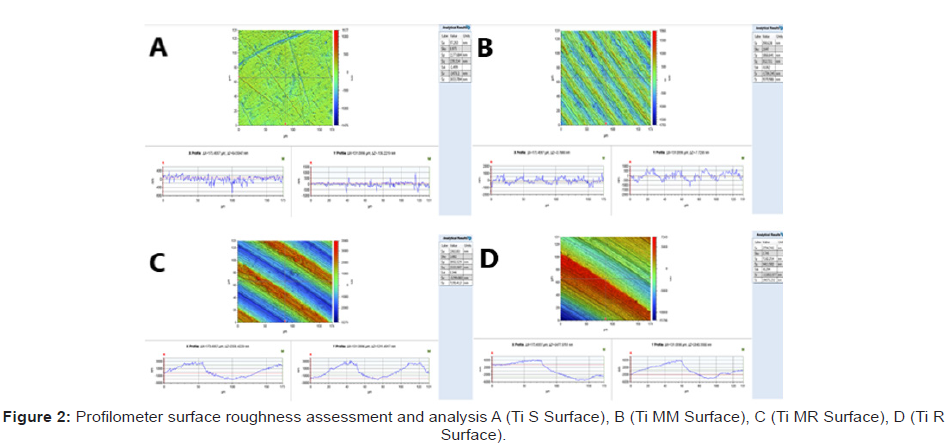
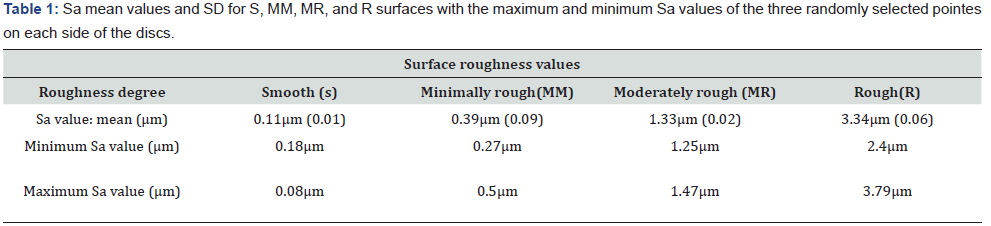
Sa values (mean – standard deviation (SD)) obtained for the S (0.11μm), MM (0.39μm), MR (1.33μm), and R (3.34μm) (Figure 2) surfaces are presented in (Table 1) below, as well as the maximum and minimum Sa values of the three randomly selected points on each disc surface.
3.2. Surface Morphology Analysis

The qualitative analysis (images) of the different roughness groups studied is illustrated in (Figure 3)
As it shows in the pictures taken by SEM (Figure 3), the differences in surface roughness of the 4 samples are clearly demonstrated. The S surface sample shows a regular slightly granular surface with some surface imperfections visible, on the top left-hand side of the image (A). The other 3 samples show concentric lines cut by the machining process, leaving a peak and trough pattern. These increase in depth and width as the surfaces go from MM (B) to MR (C) to R (D). In addition, in each sample there appears to be 2 levels of machining whereby there seems to be a further trough cut into each peak. This is not so visible in the minimally rough and moderately rough samples but is clearly visible in the rough surface.
3.3. Cell Viability
There was no statistically significant difference in the proliferation (P%) of HOB in contact with the four different roughness degrees of the Ti discs at each time point tested - day 1, 3, 5, and 10. The highest proliferation on day 1 was expressed by the cells in contact with the S surface discs (85.36%), followed by the MM surface (84.99%), then the MR surface (81.54%), and the least proliferation was expressed by the cells in contact with the R surface (79.49%). However, although the difference in P% was observed, it was not significant between all tested surfaces. At day 3, the P% dropped by almost 25% when compared to day 1 values for all the tested samples (Figure 4), and this decline in the proliferation pattern continued until day 5 where the least P% was expressed by MM surface (33.46%). S and MR surfaces showed a P% of (38.66%) and (36.69) respectively, and the maximum P% was expressed by the R surface (41.10%). At day 10, the cells expressed a P% higher than day 1 where the highest P% expressed were cells in contact with MR surface (93.96%), followed by R surface (88.63%), then MM (88.32%), with the S surface demonstrating the least P% at (87.90%) (Table 2). At day 10 all Ti surface roughness degrees illustrated comparable results in terms of HOB P% with MR surface showing the highest P% (Figure 4). The Two-way ANOVA analysis highlighted a significant time effect on cell proliferation (p < 0.0001) but no significant effect on different surface roughness degrees and no significant interaction.

3.4. Cell Cytotoxicity
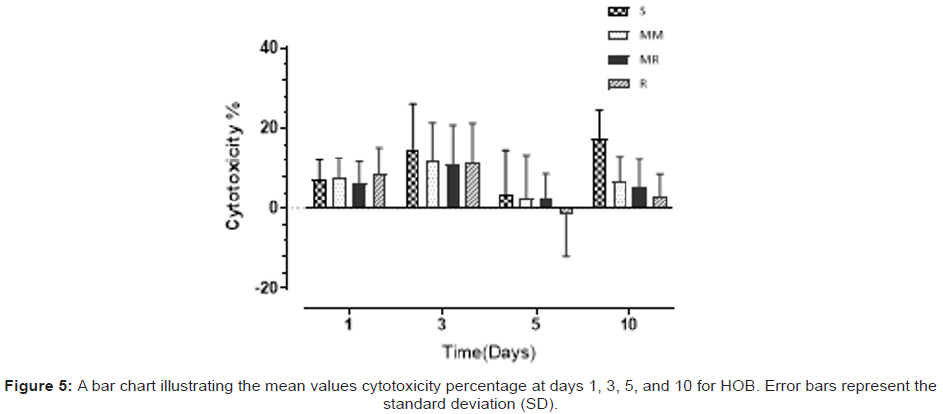
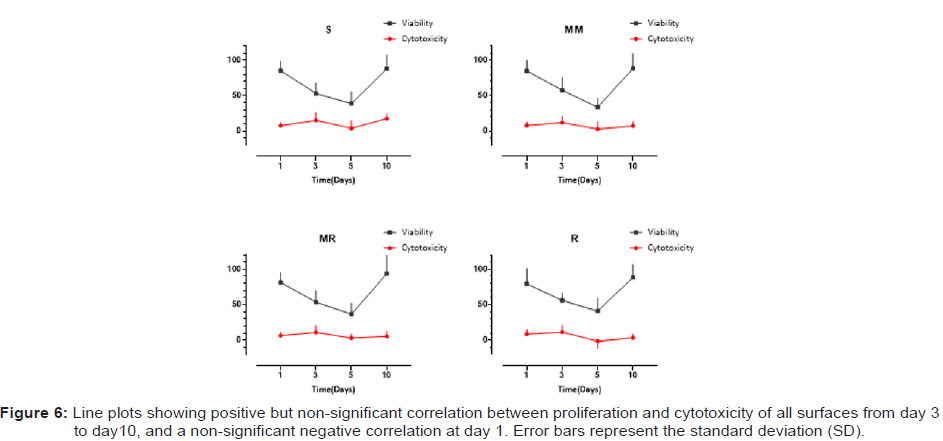
HBO cytotoxicity was generally the highest on day 3, but not statistically significant between the investigated roughness degrees (Table 3), with the following order from highest to lowest: S > MM > R > MR with cytotoxicity percentage of 14.90% > 11.74% > 11.25% > 10.94% respectively. The highest cytotoxicity % throughout the whole experiment was 17.45% at day 10 exhibited by S surface (Figure 5). In general, cytotoxicity was less than 10% at day 1, then increased on day 3 to a maximum of 14.90%, demonstrated by the S surface, then reduced to the lowest for all tested surfaces on day 5 and slightly increased again on day 10. The Two-way ANOVA analysis highlighted a significant time (p < 0.0001) and material (p < 0.03) effects on cell cytotoxicity but no significant interaction. Both assays were compared in relation to surface roughness degree over each time point (Figure 6). A nonsignificant positive correlation between viability and cytotoxicity was found for all surface roughness degrees investigated from day 3 to day 10 and a non-significant negative correlation at day 1.
4. Discussion
After the insertion of dental implants, the jawbone interacts with the implanted surface and the characteristics of that surface, such as its composition, chemistry, topography, roughness, and energy, affect the synthesis and release of local factors such as inflammatory mediators, cytokines, and growth factors produced by surrounding tissue that adhere to the implant surface including mesenchymal cells and osteoblasts. These local factors affect the process of bone formation and maturation, wound healing and eventually influence titanium biocompatibility and osseointegration/contact with bone [35]. The surface composition, roughness and topography can greatly affect the proliferation and protein synthesis of osteoblast cells that are cultured on a metal substrate [35], and this may have an effect on bony growth, especially in the early phase of healing.
Numerous techniques have been developed during the last 50 years with the aim of improving bone-to-implant contact and accelerating osseointegration from a physical and/or chemical perspective [36]. The first Osseo integrated implant surfaces were produced by industrial machining technique, which led to minimally rough surfaces with some residual periodic microgrooves. Despite the clinical success of these machined surfaces, further processes have been developed to improve the microtopography and surface roughness in order to achieve larger surface area which consequently led to better bone-toimplant contact and faster osseointegration, using either additive or subtractive methods. These methods include, but are not limited to, titanium plasma spraying, acid-etching, grit-blasting, anodization, laser, antimicrobial, and growth factor coating [37]. However, many of these developments have been driven by clinical observations and not quantified at a cellular level through “in-vitro” investigations.
In the present study, four different Ti surface roughness degrees with the same preparation technique (industrial machining) were investigated in terms of their influence on HOB’s proliferation and cytotoxicity. The present study demonstrated no statistically significant difference in HOB cell proliferation between all the investigated roughness degrees, thus, the first null hypothesis was accepted. However, the Two-way ANOVA analysis highlighted a significant time effect on cell proliferation (p < 0.0001). In term of cytotoxicity, there was no statistically significant difference in the toxic effect between all the tested surfaces when HOB cells were exposed to them. However, on day 10, S surface showed the highest % of HBO cytotoxicity throughout the whole experiment (17.45%) which was more than double the other surfaces’ figures, and the Two-way ANOVA analysis highlighted a significant time (p < 0.0001) and surface (p < 0.03) effect on cell cytotoxicity. However, no significant interaction was found, thus indicating…... Additionally, the 30% cut off percentage of cytotoxicity was not reached by any of the surfaces at any time point which implies that all surfaces were biocompatible and non-toxic. These results mean, the second null hypothesis was partially accepted. Furthermore, according to the analysis of our results, there were also significant time effects on both cell proliferation (p < 0.0001) and cytotoxicity (p < 0.0001), and consequently, the third hypothesis was rejected.
Different surface roughness degrees result in discrete effects on different living tissues and cells [38]. On a three-dimensional (3-D) scale, smooth surface was defined as having an average height deviation (Sa) of < 0.5μm, minimally rough surfaces Sa 0.5 - 1μm, moderately rough surfaces Sa 1- 2μm, and rough surfaces Sa > 2μm which are the values that were used in this study [39,40]. Moderately rough and rough surface roughness degrees are associated with stronger bone responses, meaning enhanced proliferation percentage and less cytotoxic effects than the smooth and minimally rough surfaces noticed in this study. These results are in line with several other in-vitro and in-vivo studies [41,42], and a potential optimal range of roughness for implant osseointegration in terms of the moderately rough surface [43, 44]. At a cellular level, although it was not investigated in this study moderate roughness may be more optimal for cell attachment, while the rough surface leaves longer distances between the peaks of the surface that bone cells perceive them as flat, causing excessive cells flattening and compromising their attachment and nutrition, so that, it affects their attachment and proliferation [27].
The impact of macro-roughness on dental implants is primarily mechanical, in which, the surface irregularities mechanically strengthening the implant anchorage withing the jawbone but being too large to be influential on cells [41]. In contrast, microand nano-topographies (surface roughness) appear to influence osteogenesis and cellular behaviour through alterations in mesenchymal stem cell biological function [38]. The surface chemistry, energy, wettability, and the mechanical properties of the commercially available dental implants might also be different compared to the fabricated Ti surfaces used in this experiment [45], however, these properties were not of interest in this study as it was purely the biocompatibility of different surface roughness degrees (created by simple industrial milling machine) of the exact same material under static biological conditions, and the quantification of its effect on HBO’s that was being investigated. Expectedly, as it has been reported in many studies [45], an increase in the surface roughness leads to an increase in the total surface area of the implant. This means a larger contact area for the attachment of cells is present, aa well as the binding of important and relevant proteins, inflammatory mediators, and growth factors. This was the case in this experiment where MR and R surfaces exhibited more proliferation percentage and much less cytotoxicity than the S and MM surfaces. This finding is consistent with several previous studies which also reported that increasing degrees of Ti surface roughness elicit enhanced levels of bone cell proliferation in vitro [46,47]. In contrast, several authors corroborate the present results, observing greater cell proliferation on the Ti surface as roughness decreases [48-51].
In the present experiment, cell proliferation decreased at day 3 and 5, and then dramatically increased at day 10. In part, this drop in cell proliferation could be due to the short time interval (2days), which technically allows less time for the cells to acclimatize and proliferate, but toward day 10, where the cells have a 5-day interval, the proliferation level increases again. This decrease in cell proliferation should not necessarily imply that cells are dying, as they might be reacting to the surface by differentiation rather than proliferation, and increased proliferation might be a reaction to environmental stimuli [32, 52]. The role of the surface roughness on HOB cell behaviour and metabolic activity has been demonstrated in previous in-vitro and in-vivo studies [53, 54]. Further findings were reported by Deligianni et al. after investigating the influence of three different roughnesses of hydroxyapatite (HA) surfaces (smooth, machined, and rough) fabricated via grinding papers on HA coated Ti discs [55]. The roughness of HA Ti surface had no impact on bone marrow stromal cells morphology or ALP activity. However, cellular proliferation after 14 days was higher on coarser HA when compared to smoother surfaces [55], and same observation was illustrated by Kunzler et al. where they reported a significant increase in the number and proliferation rate of rat osteoblasts by increasing the surface roughness gradients of high purity aluminium [56]. The increased proliferation results on rougher surfaces are comparable to the results in our study results with primary HOB cells after ten days in-vitro with machine created roughness. A period of 10 days may be too short to detect significant differences in the proliferation of HOB on Ti surface, however it does indicate that early population of HOB on Ti surfaces may be enhanced by increased roughness.
All the investigated roughness degrees showed comparable cytotoxicity levels with the S surface marked as the highest at day 10 with 17.45%, however, none of which exceeded the ISO standard level of cytotoxic material (30%) [57]. Therefore, this implies that all the surfaces investigated may be suitable invivo use, and that other characteristics my influence short term healing and osseointegration. Each disc surface area was 223mm² which is within the range of the surface area of a 11mm x 4mm Ø and 13mm x 4.5mm Ø dental implant, which is representative of the clinical indications of the investigated surfaces, in that they were comparable to those surface areas of routinely used dental implants. Additionally, the Ti discs were incubated after cells seeding at 37°C to simulate oral conditions [57]. Furthermore, the ratio of the surface area of the sample to medium volume was 3 cm2/mL, which is within the ISO standard ratio of 0.5−6 cm2/ mL, for such investigations, ISO 10933, 12 [58]. A final point to highlight was that an HOB primary cell line was used in these investigations rather than immortalised or carcinoma cells, which have been used in other studies, as it was felt they were more representative of healthy human cells located within the oral environment [59,60], in direct contact with the Ti surfaces, which was considered the most clinically comparable and sensitive method to measure low cytotoxicity levels [61].
5. Conclusion
In these investigations, we studied the influence of different surface roughness degrees created by industrial machining (milling only with no other surface treatments) on HOB proliferation and cytotoxicity. Applying only a simple surface treatment technique allowed the effects of the surface roughness to be isolated from differences in surface chemistry, crystal structure, surface energy and relative density that was shown when comparing same roughness degrees with different techniques of making them. The changes of HOB proliferation on the studied surfaces after 10 days were not significant (p > 0.05) between all groups, evaluated by two-way ANOVA and Tukey’s Multiple Comparison Method. All investigated roughness degrees showed comparable HOB proliferation with MR surface presenting the highest percentage followed by R, MM, and S, respectively. S surface showed the highest inhibitory effect on HOB; however, it did not reach the cytotoxic level as suggested by ISO for any medical device to be considered cytotoxic. While a rough surface may increase boneto- implant contact, it can also induce inflammatory cytokine production, which affects bone resorption [62]. Therefore, careful consideration is required in implant design or different treatment methods may be needed to increase the surface roughness, as too much roughness could have adverse effects on the cellular environment and dramatically increase the production of inflammatory cytokines from the host tissue as well as bacterial and debris adhesion, leading to soft tissue inflammation, infection, alveolar bone resorption and eventually, implant loss. Finally, it is important to highlight that simple mailing procedure to create surface roughness can produce – to a certain extent – similar/comparable biological effect on HOB to other more expensive and complicated procedures and techniques.
References
- Adell R, Hansson BO, Brånemark PI, Breine U (1970) Intra-osseous anchorage of dental prostheses. II. Review of clinical approaches. Scand J Plast Reconstr Surg 4(1): 19-34.
- Albertsson T, Wennerberg A (2005) The impact of oral implants - past and future, 1966-2042. J Can Dent Assoc 71(5): 327.
- Branemark PI, Adell R, Breine U, Hansson BO, Lindström L, et al. (1969) Intra-osseous anchorage of dental prostheses. I. Experimental studies. Scand J Plast Reconstr Surg 3(2): 81-100.
- Branemark PI, Adell R, Breine U, Hansson BO, Hellen O, et al. (1977) Osseointegrated implants in the treatment of the edentulous jaw. Experience from a 10-year period. Scand J Plast Reconstr Surg Suppl 16: 1-132.
- Rupp F, Liang L, Geis Gerstorfer J, Scheideler L, Hüttig F (2018) Surface characteristics of dental implants: A review. Dent Mater 34(1): 40-57.
- Thomsen P, Larsson C, Ericson LE, Sennerby L, Lausmaa J, et al. (1997) Structure of the interface between rabbit cortical bone and implants of gold, zirconium and titanium. J Mater Sci Mater Med 8(11): 653-665.
- Brunette DM, Pentti Tengvall, Marcus Textor, Peter Thomsen, et al. (2001) Titanium in medicine: material science, surface science, engineering, biological responses and medical applications.
- Sola-Ruiz MF, Perez-Martinez C, Rueda CL, Carda C, Javier Martín De Llano J (2017) Behavior of Human Osteoblast Cells Cultured on Titanium Discs in Relation to Surface Roughness and Presence of Melatonin. Int J Mol Sci 18(4): 823.
- Albrektsson T, Hansson BO, Brånemark PI, Lindström L (1981) Osseointegrated titanium implants. Requirements for ensuring a long-lasting, direct bone-to-implant anchorage in man. Acta Orthop Scand 52(2): 155-170.
- Ananth H, Vinaya Kundapur, Mohammed HS, Anand M, Amarnath GS, et al. (2015) A Review on Biomaterials in Dental Implantology. Int J Biomed Sci 11(3): 113-120.
- Li Y, Yang C, Zhao H, Shengguan Qu, Xiaoqiang Li, et al (2014) New Developments of Ti-Based Alloys for Biomedical Applications. Materials (Basel) 7(3): 1709-1800.
- Saini M, Yashpal Singh, Pooja Arora, Vipin Arora, Krati Jain (2015) Implant biomaterials: A comprehensive review. World J Clin Cases 3(1): 52-57.
- Civantos A (2017) Titanium Coatings and Surface Modifications: Toward Clinically Useful Bioactive Implants. ACS Biomater Sci Eng 3(7): 1245-1261.
- Bostman O, Hirvensalo E, Mäkinen J, Rokkanen P (1990) Foreign-body reactions to fracture fixation implants of biodegradable synthetic polymers. J Bone Joint Surg Br 72(4): 592-596.
- Henkel J, Woodruff MA, Epari DR, Steck R, Glatt V, et al (2013) Bone Regeneration Based on Tissue Engineering Conceptions - A 21st Century Perspective. Bone Res 1(3): 216-248.
- Hench LL (2006) The story of Bioglass. J Mater Sci Mater Med 17(11): 967-978.
- Hench LL, Polak JM (2002) Third-generation biomedical materials. Science 295(5557): 1014-1017.
- Asri RI, Harun WSW, Samykano M, Lah NAC, Ghani SAC, et al. (2017) Corrosion and surface modification on biocompatible metals: A review. Mater Sci Eng C Mater Biol Appl 77: 1261-1274.
- Neoh KG, Hu X, Zheng D, Kang ET (2012) Balancing osteoblast functions and bacterial adhesion on functionalized titanium surfaces. Biomaterials 33(10): 2813-2822.
- Boyan BD, Lossdörfer S, Wang L, Zhao G, Lohmann CH, et al. (2003) Osteoblasts generate an osteogenic microenvironment when grown on surfaces with rough microtopographies. Eur Cell Mater 6: 22-27.
- Schwartz Z, Raz P, Zhao G, Barak Y, Tauber M, et al. (2008) Effect of micrometer-scale roughness of the surface of Ti6Al4V pedicle screws in vitro and in vivo. J Bone Joint Surg Am 90(11): 2485-2798.
- De Bruyn H, Christiaens V, Doornewaard R, Jacobsson M, Cosyn J, et al. (2017) Implant surface roughness and patient factors on long-term peri-implant bone loss. Periodontol 2000 73(1): 218-227.
- Cochran DL (1999) A comparison of endosseous dental implant surfaces. J Periodontol 70(12): 1523-1539.
- Larsson L, Pilipchuk SP, Giannobile WV, Castilho RM, (2018) When epigenetics meets bioengineering-A material characteristics and surface topography perspective. J Biomed Mater Res B Appl Biomater 106(5): 2065-2071.
- Fickl S, Kebschull M, Calvo-Guirado JL, Hürzeler M, Zuhr O (2015) Experimental Peri-Implantitis around Different Types of Implants - A Clinical and Radiographic Study in Dogs. Clin Implant Dent Relat Res 17(2): e661-669.
- Stepanovska J, Matejka R, Rosina J, Bacakova L, Kolarova H, et al. (2020) Treatments for enhancing the biocompatibility of titanium implants. Biomed Pap Med Fac Univ Palacky Olomouc Czech Repub 164(1): 23-33.
- Wennerberg A, Albrektsson T (2009) Effects of titanium surface topography on bone integration: a systematic review. Clin Oral Implants Res 20(4): 172-184.
- Albrektsson T, Wennerberg A (2004) Oral implant surfaces: Part 1 review focusing on topographic and chemical properties of different surfaces and in vivo responses to them. Int J Prosthodont 17(5): 536-543.
- Zitzmann NU, Berglundh T (2008) Definition and prevalence of peri-implant diseases. J Clin Periodontol 35(8): 286-291.
- Rupp F, Liang L, Geis-Gerstorfer J, Scheideler L, Hüttig F (2018) Surface characteristics of dental implants: A review. Dental Materials 34(1): 40-57.
- Rupp F, Scheideler L, Rehbein D, Axmann D, Geis-Gerstorfer J (2004) Roughness induced dynamic changes of wettability of acid etched titanium implant modifications. Biomaterials 25(7-8): 1429-1438.
- Alamoush RA, Kushnerev E, Yates JM, Satterthwaite JD, Silikas N (2020) Response of two gingival cell lines to CAD/CAM composite blocks. Dent Mater 36(9): 1214-1225.
- Scientific TF (2018) Thermo Fisher Scientific. Available at: alamarBlue® Thermo Fisher Scientific Inc.
- Scientific TF (2014) Thermo Fisher Scientific. Available at: Pierce LDHcytotoxicity assay kit. Thermo Fisher Scientific.
- Kieswetter K, Schwartz Z, Dean DD, Boyan BD (1996) The role of implant surface characteristics in the healing of bone. Crit Rev Oral Biol Med 7(4): 329-345.
- Coelho PG, Granjeiro JM, Romanos GE, Suzuki M, Silva NR, et al. (2009) Basic research methods and current trends of dental implant surfaces. J Biomed Mater Res B Appl Biomater 88(2): 579-596.
- Dohan Ehrenfest DM, Coelho PG, Kang BS, Sul YT, Albrektsson T (2010) Classification of osseointegrated implant surfaces: materials, chemistry and topography. Trends Biotechnol 28(4): 198-206.
- Ellingsen JE (1998) Surface configurations of dental implants. Periodontol 2000 17: 36-46.
- Goriainov V, Cook R, Latham MJ, Dunlop GD, Oreffo RO (2014) Bone and metal: an orthopaedic perspective on osseointegration of metals. Acta Biomater 10(10): 4043-4057.
- Wennerberg A, Albrektsson T (2000) Suggested guidelines for the topographic evaluation of implant surfaces. Int J Oral Maxillofac Implants 15(3): 331-344.
- Bacakova L, Filova E, Parizek M, Ruml T, Svorcik V (2011) Modulation of cell adhesion, proliferation and differentiation on materials designed for body implants. Biotechnol Adv 29(6): 739-767.
- Anselme K, Bigerelle M, Noël B, Iost A, Hardouin P (2002) Effect of grooved titanium substratum on human osteoblastic cell growth. J Biomed Mater Res 60(4): 529-540.
- Ronold HJ, Lyngstadaas SP, Ellingsen JE (2003) Analysing the optimal value for titanium implant roughness in bone attachment using a tensile test. Biomaterials 24(25): 4559-4564.
- Yang A, et al (2017) Optimum surface roughness prediction for titanium alloy by adopting response surface methodology. Results in Physics 7: 1046-1050.
- Holthaus MG, Treccani L, Rezwan K (2013) Osteoblast viability on hydroxyapatite with well-adjusted submicron and micron surface roughness as monitored by the proliferation reagent WST-1. J Biomater Appl 27(7): 791-800.
- Brett PM, Harle J, Salih V, Mihoc R, Jones FH , et al (2004) Roughness response genes in osteoblasts. Bone 35(1): 124-133.
- Degasne I, Baslé MF, Demais V, Huré G, Lesourd M, et al. (1999) Effects of roughness, fibronectin and vitronectin on attachment, spreading, and proliferation of human osteoblast-like cells (Saos-2) on titanium surfaces. Calcif Tissue Int 64(6): 499-507.
- Balloni S, Calvi EM, Damiani F, Bistoni G, Calvitti M, et al. (2009) Effects of titanium surface roughness on mesenchymal stem cell commitment and differentiation signaling. Int J Oral Maxillofac Implants 24(4): 627-635.
- Gittens RA, McLachlan T, Olivares-Navarrete R, Cai Y, Berner S, et al. (2011) The effects of combined micron-/submicron-scale surface roughness and nanoscale features on cell proliferation and differentiation. Biomaterials 32(13): 3395-3403.
- Kim MJ, Kim CW, Lim YJ, Heo SJ (2006) Microrough titanium surface affects biologic response in MG63 osteoblast-like cells. J Biomed Mater Res A 79(4): 1023-1032.
- Olivares-Navarrete R, Gittens RA, Schneider JM, Hyzy SL, Haithcock DA, et al. (2012) Osteoblasts exhibit a more differentiated phenotype and increased bone morphogenetic protein production on titanium alloy substrates than on poly-ether-ether-ketone. Spine J 12(3): 265-272.
- Lloyd AC (2013) The regulation of cell size. Cell 154(6): 1194-1205.
- Ball MD, Downes S, Scotchford CA, Antonov EN, Bagratashvili VN, et al. (2001) Osteoblast growth on titanium foils coated with hydroxyapatite by pulsed laser ablation. Biomaterials 22(4): 337-347.
- Hayashi K, Inadome T, Tsumura H, Nakashima Y, Sugioka Y (1994) Effect of surface roughness of hydroxyapatite-coated titanium on the bone-implant interface shear strength. Biomaterials 15(14): 1187-1191.
- Deligianni DD, Katsala ND, Koutsoukos PG, Missirlis YF (2001) Effect of surface roughness of hydroxyapatite on human bone marrow cell adhesion, proliferation, differentiation, and detachment strength. Biomaterials 22(1): 87-96.
- Kunzler TP, Drobek T, Schuler M, Spencer ND (2007) Systematic study of osteoblast and fibroblast response to roughness by means of surface-morphology gradients. Biomaterials 28(13): 2175-2182.
- Iso B, Standard B (2009) Biological evaluation of medical devices. Part 1: 10993.
- Standardization IOf (2007) Biological Evaluation of Medical Devices: Sample Preparation and Reference Materials. ISO.
- Pabst A, Walter C, Bell A, Weyhrauch M, Schmidtmann I , et al. (2016) Influence of CAD/CAM zirconia for implant-abutment manufacturing on gingival fibroblasts and oral keratinocytes. Clinical oral investigations 20(5): 1101-1108.
- Pendegrass C, Lancashire HT, Fontaine C, Chan G, Hosseini P, et al. (2015) Intraosseous transcutaneous amputation prostheses vs dental implants: A comparison between keratinocytes and gingival cell adhesion in vitro. European Cells and Materials 29: 237-249.
- De Melo WM, Amarú Maximiano WM, Antunes AA, Beloti MM, Rosa AL, et al. (2013) Cytotoxicity testing of methyl and ethyl 2-cyanoacrylate using direct contact assay on osteoblast cell cultures. Journal of Oral and Maxillofacial Surgery 71(1): 35-41.
- Naganawa T, Ishihara Y, Iwata T, Koide M, Ohguchi M, et al. (2004) In vitro biocompatibility of a new titanium-29niobium-13tantalum-4.6zirconium alloy with osteoblast-like MG63 cells. J Periodontol 75(12): 1701-1707.






























