Retrospective Coherent Study of Immediate Effect of Blue Light on the Dental Pulp
Samir Koheil* and Yousriya Shalaby
Professor of Conservative Dentistry, Faculty of Dentistry, Alexandria University, Egypt
Submission: March 29, 2021; Published: April 08, 2021
*Corresponding author: Samir Koheil, Professor of conservative Dentistry, Faculty of Dentistry, Alexandria University, Egypt
How to cite this article: Samir K, Yousriya S. Retrospective Coherent Study of Immediate Effect of Blue Light on the Dental Pulp. Adv Dent & Oral Health. 2021; 14(1): 555878. DOI:10.19080/ADOH.2021.14.55878
Abstract
Statement of problem: This retrospective coherent study was carried out to study the effect of blue light on the dental pulp during the composite curing.
Purpose: Histological and histochemical examination of dental pulp of non caries teeth extracted for orthodontic treatment of young age patients.
Material and methods: The present study included 18 sound teeth indicated for extraction for orthodontic treatment. Class v prepared in all the teeth, 14 filled with light-cured composite, and four left unfilled (control). All 18 teeth were then extracted individually. Its pulps were investigated histologically by H & E and histo chemically for alkaline Phosphatase, acid Phosphatase, and succinic dehydrogenase.
Results: The results were as follows: Increased alkaline Phosphatase, acid Phosphatase, and succinic dehydrogenase activity were shown by all the pulp tissue except a decrease alkaline phosphatase reaction blood avascular wall of the pulp of teeth exposed to light. The increased enzymatic reaction in the pulp tissue with fibroblastic hyperplasia and vasodilation supported the irritatory and not inflammatory effect on the dental pulp.
Conclusion: The blue light has a definite histological and injurious histochemical effect on the pulp tissue. This histochemical effect demonstrates the expected harmful effect that will be demonstrated histologically later on.
Keywords: Pulp tissue; Retina; Senile macular; Alkaline Phosphatase; Acid Phosphatase; Odontoblastic layer; Control specimen
Introduction
Visible light sources have a broad peak in the visible range between 400 and 600 nm. Even though visible light higher in the spectrum and is therefore considered safer for the eyes. Many clinicians experience showed after image or eye strain when using these light units that light with a wavelength of less than 500 nm may contribute to the premature aging of the retina and too senile macular degeneration (the decreasing ability of the macular region of the retina and too senile macular region of the retina to provide visual acuity) [1,2]. Different light-cured units, despite being on the low end of the potential harm or the high end of discomfort, are sufficient to at least impair visual performance for a short period or perhaps even to inflict permanent ocular damage [3]. The council of dental materials (1985) [4] stated that operators working with the light-curing unit should wear a protective filtering device, eyeglasses, or eye shield while curing light-activated resin composite. Heat omitted during the cure of the composite end with the hypothesis that visible light cure lamps may cause a temperature increase in the pulp chamber that harms the dental pulp itself [5]. Applied enzyme histochemistry is a useful technique which can be used to elucidate changes occurred in cell organelles when expected harmful effect on the pulp cells pathological conditions. It also elucidates normal physiological and biochemical activities that undergo in normal tissues. As such, these changes could not be perceived in routine histopathological studies [6]. It is noticed that some cases complained of post-operative sensitivity of teeth after filling with light-cured composite [7] .
Aim of the Work
The aim was to study the reactivity and organization of the connective tissue of the dental pulp and the changes of the tissue colloids during the biological effect of blue light of the light cure units used polymerize composite resin filling material.
Material and Methods
Eighteen sound upper premolars were selected for this
study from the orthodontic department, indicated for immediate
extraction, Faculty of Dentistry, Alexandria University. Class V
cavities were prepared in all the teeth’ dentin provided the gingival
wall 1mm occlusal to the cervical line. Mesially and distally, the
cavities extended to the axial line angles. Fourteen of these teeth
were filled with light-cured composite following the etch and rinse
total-etch technique. The composite was applied on the mesial part
of Class V and cured for 40 seconds and then on the distal part and
cured for 40 seconds. The whole cavity was filled, contoured, and
cured for 40 seconds. The composite filling was over cured for 20
seconds. Each tooth subjected to the blue intensified light was 140
seconds sequentially. The remaining four teeth are not filled and
considered as control where teeth are prepared but not filled. The
eighteen filled and unfilled teeth were extracted, and the pulps of
these teeth were subjected to the following classification:
a) Group I: Four pulps of the teeth that were prepared, not
filled, and not subjected to light were divided into.
i. Subgroup A: Pulps of two teeth of Group I were subjected
to histological examination.
ii. Subgroup B: Pulps of the other two teeth of Group II
were subjected to histochemical examination.
b) Group II: Fourteen pulps of the teeth that were prepared,
filled with composite resin, and light-cured were divided into.
i. Subgroup C: Seven pulps of Group I was subjected to
histological examination.
ii. Subgroup D: The seven remaining pulps of Group II
were subjected to histochemical examination.
The histological and histochemical techniques that were
applied as follows:
Histological study
The pulp of the two pulps of (subgroup A) and the seven pulps
of (subgroup II C) were carefully detached and fixed in 10% formal
saline and processed to get 6-micron thick paraffin sections for
the histological study where haematoxyline and eosin stain was
used.
Histochemical study
The pulp of the two teeth of (subgroup B) and seven teeth of
subgroup D were carefully detached, immediately frozen, and cut
by cryostat into 10-micron thick sections.
Frozen sections were incubated in each appropriate substrate
for the following enzymes:
a) Subgroup B 1- Alkaline Phosphatase.
b) Subgroup B 2-Acid Phosphatase
c) Subgroup B 3-Succinic Dehydrogenase
i. Subgroup D 1- Alkaline Phosphatase
ii. Subgroup D 2-Acid Phosphatase
iii. Subgroup D 3-Succinic Dehydrogenase
Antifungal resistance
Results
Histological results
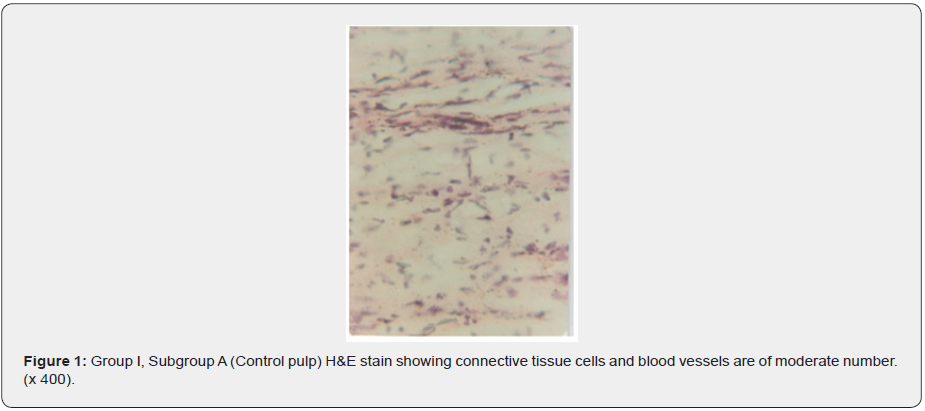
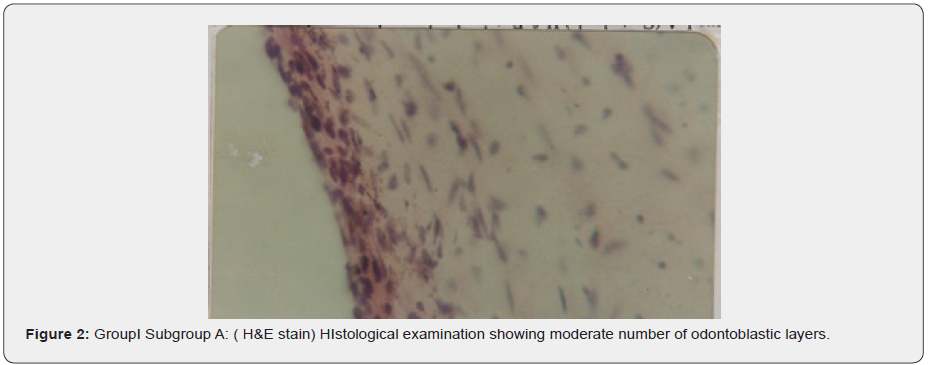
a) Subgroup A: The histological pulps tissue that was not subjected to light showed a moderate number of connective tissue cells (mainly fibroblast) and a small number of blood vessels (Figure 1). While the odontoblastic layer showed a moderate number of cellular layers (Figure 2).
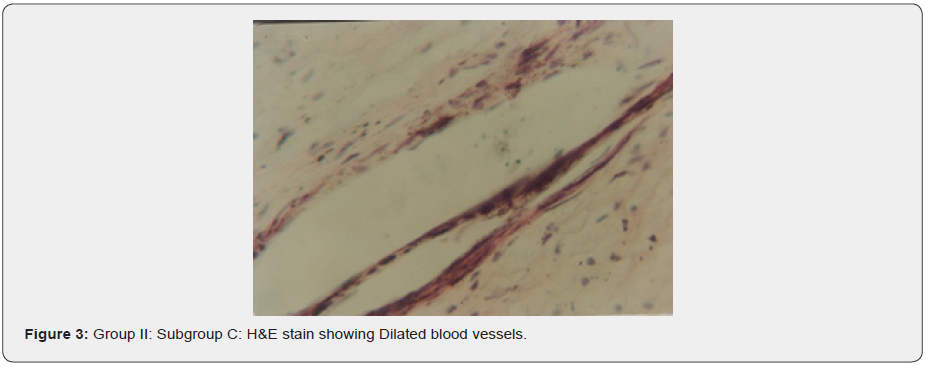
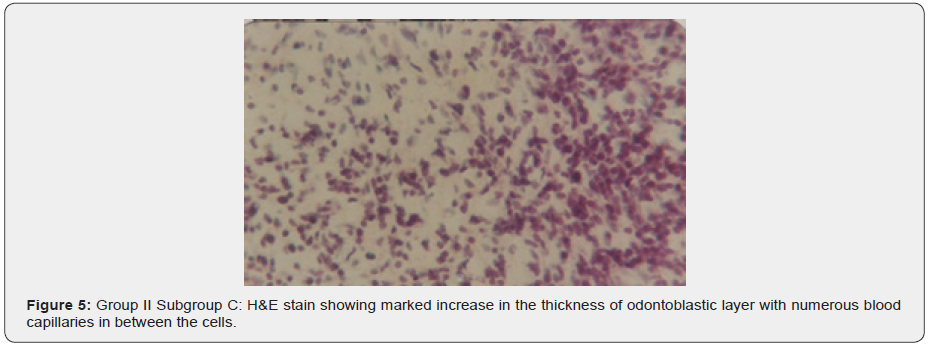
b) Subgroup C: The histological pulp tissue of the seven specimens revealed evident dilatation of blood vessels (Figure 3) with an increase in the number of connective tissue cells, mainly fibroblast (Figure 4). The odontoblastic layer showed an increase in its thickness and numerous blood capillaries between its cells (Figure 5).

Histochemical results
Alkaline phosphatase
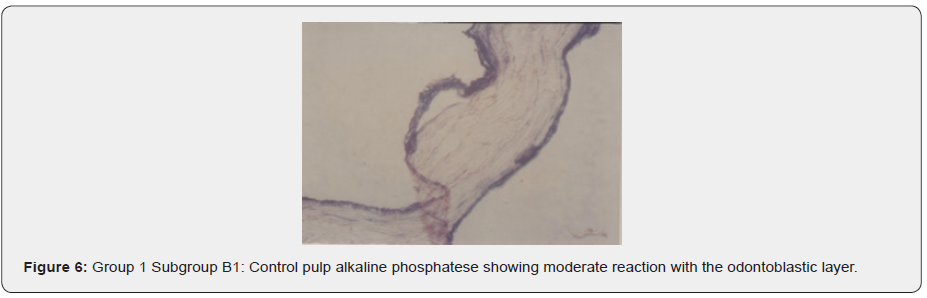
a) Subgroup B1: (Control specimen) When incubated in
alkaline Phosphatase, the blood capillaries’ endothelial lining
showed moderate enzymatic activity. The thin odontoblastic layer
showed an intense reaction, and some of the connective tissue
cells showed a muted reaction (Figure 6 & 7).
b) Subgroup D1: When incubated in alkaline Phosphatase, the blood endothelial cells lining of the blood capillaries showed decreased enzymatic activity. A strong and heavy enzymatic reaction of the odontoblastic layer was detected. The increased reaction was noticed in the fibroblast (Figure 8).


Acid phosphatase
a) Subgroup B 2. That incubated in acid phosphatase, pulps
showed faint acid phosphatase reaction in the pulp’s connective
tissue, but the odontoblastic layer showed moderate enzymatic
activity (Figure 9).
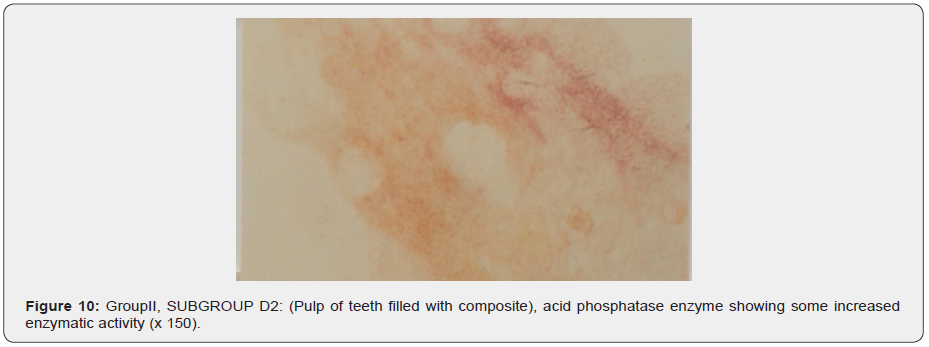
b) Subgroup D2. That incubated in acid phosphatase, pulps
showed some increase in acid phosphatase activity (Figure 10).

Succinic dehydrogenase
a) Subgroup B 3: That incubated in succinic dehydrogenase
showing moderate succinic dehydrogenase activity in the
odontoblastic layer and fainter in the connective tissue cells,
mainly fibroblast (Figure 11).
b) Subgroup D 3: That incubated in succinic dehydrogenase
showing an increase in succinic dehydrogenase enzyme activity
mainly in the odontoblastic layer and the fibroblast of the
connective tissue (Figure 12).


Discussion
This study was carried out to study blue intensified fixed light on the dental pulp through exposure to light when filling class V cavities with light-cured composite. Cavities were prepared for the patient seeking orthodontic treatment, and that was indicated for extraction; cavities were filled with light-cured composite. All the precautions were carried out to no trauma transmitted to the pulp during cavity preparation. The work was divided into two parts, one with only cavity preparation and without filled with composite resin material, and the other filled with a composite resin filling material and so the difference was the result of light effect on the dental pulp. Each tooth was exposed to 140 seconds of blue light for polymerization of composite “simulating filling of the large cavity.” The selected teeth were extracted. The pulps of teeth were detached and prepared for histological and histochemical study. Histologically, the present result showed; the specimens exposed to an intensified light increase in thickness in the odontoblastic layer (hyperplasia) and the fibroblast in the pulp’s connective tissue were increased in number, and lastly, vasodilatation of blood vessels was evident.
This agreed with Zhi-Chun Zhao [2]; Fumihiko Yoshino &
Ayaka Yoshida et al. [8] who stated that light pollution influences their eyes. In the visible spectrum, short-wave blue light with a
wavelength between 415 nm and 455 nm is closely related to
eye light damage. This high-energy blue light passes through
the cornea and lens to the retina, causing diseases such as dry
eye, cataract, age-related macular degeneration. Furthermore,
Isabella [9] stated that Immediate and excessive superficial wear
of a recently placed resin composite generates alterations in the
resin matrix by the heat produced, disturbs the post-irradiation
phase polymerization, and removes the superficial layer, which
theoretically obtains the highest degree of conversion. Also,
Mamalis A et al. [10] investigated the effects of LED-Blue light
on human skin fibroblast proliferation. Also, Ieda N et al. [11];
Yoshida A et al. [12] oxidative stress-induced hyperfunction in oral
mucosal cells. In particular, the blue-light irradiation of gingival
fibroblasts increases. Yoshida A [13] agreed and demonstrated
temporally controlled vasodilation of rat aorta ex vivo by bluelight.
Histochemically, the present result showed that the pulp
demonstrated an increase in the alkaline phosphatase activity
in both the odontoblastic and fibroblast in the connective tissue
where there was increased reaction reactions odontoblast of the
connective tissue of the treated pulp. This agreed with Martin S et
al. [14]; Zhu T et al. [15], who stated that the results of The levels
of alkaline Phosphatase with the groups were irradiated with blue
light were higher in every experimental group than in the control
group. Noting the involvement of tissue-nonspecific alkaline
phosphatase increased reaction of the alkaline Phosphatase might
lead to a fibroblastic activity. Also, Abe T et al. [16]. Who found that
wound healing and inflammation, fibroblasts express elevated
alkaline Phosphatase; they hypothesized that the extracellular
matrix environment might influence the induction of alkaline
Phosphatase in fibroblasts who stated that ophthalmic research
into the appearance of the blue light lesion has not ruled out the
possibility that intense wavelength light (less than 500nm) may
contribute to the premature aging of the retina and too senile
macular degeneration and blue light also may cause the formation
of cataract (fibrous degeneration of the lens).
Simultaneously, the pulps’ blood capillaries showed dilatation
and an evident decrease in alkaline phosphatase activity of the
lining endothelium. This might be a result of the reflection of the
injury of the endothelial cells. While Alonso LRJ et al. [17]. Agreed
and found that the blood capillaries of pulps showed dilatation
and an evident decrease in alkaline phosphatase activity of the
lining endothelium. This might be a result of the reflection of the
injury of the endothelial cells. This also agreed with Perticone F et
al. [18], who stated a significant and robust inverse relationship
between alkaline phosphatase levels and endothelium-dependent
vasodilation. Also agreed with Fumihiko Yoshino F & Yoshida A
[19]. Dentists should be aware that the radiation can cause various
phototoxic and photoallergic reactions. The result showed that
acid phosphatase increased in the pulp tissue activity, including
the odontoblastic layer and the connective tissue; this might
represent an injury to the pulp itself, leading to its irritation. A
reaction from the part of the tissue is to cope with the product of
the injury.
This agreed with Suter A [20], Who stated that acid
phosphatase is a lysosomal enzyme. Moreover, H Bull et al. [21]
found that identity and diversity of acid Phosphatase and the
relation between acid Phosphatase and the phagocytic mechanism
of the cells and human disease and clinical diagnosis. The present
result showed increased activity in the succinic dehydrogenase;
this might be due to mitochondrial irritation by the blue light,
which might lead to multiplication and increased number of
mitochrondial or their rupture resulting in releasing the succinic
enzyme dehydrogenase free in the cells. This agreed with Tian P
[22], who stated that Succinate dehydrogenase is a mitochondrial
marker enzyme. Furthermore, Rutter J [23] stated that it plays an
essential role in cell metabolism. The present study showed that
the histological and histochemical picture is a sort of irritation to
pulp cells, which might be due to the effect of the energy elicited
by the wavelength used in blue light. This agrees with Wu J et al.
[24], who stated that light induced retinal damage can be hastened
by increased exposure to visible light with a wavelength of less
than 500 nm. This effect is photochemical rather than thermal or
structural.
In this study, the teeth selected were sound of young age with
healthy pulp and when exposed to blue light showed an irritating
effect on the pulp tissue quite different from that of infection. In
the first, the studied three enzymes’ activity showed an evident
increase in the pulp tissue except in the vascular blood wall,
compared to the second where alkaline Phosphatase, especially
in the vascular wall, showed an evident increase. In the present
study, the fibroblast showed an evident increase in number
while in infection, mononuclear and polympnuclear cells are the
predominant ones. The result showed increased thickness of
the odontoblastic layer, which increased as a result of irritation,
which, when stimulated, will lead to the formation of secondary
dentin to protect the pulp. The present study demonstrated both
histological and histochemical findings. The histochemical results
were more manifested and supported the histological results as
it appears on subcellular changes. One can expect histological
changes in the pulp tissue after a long time compared to the
histochemical findings. Subcellular level showing the early cellular
changes. One could expect histological changes in the pulp tissue
after a long time compared to the histochemical findings.
Conclusion
The blue light has a definite histological and injurious histochemical effect on the pulp tissue. The histochemical findings showed the earliest change of cells before demonstrated histologically. It is expected that increased reaction of enzymes in the odontoblastic layer might lead to increased fibroblastic activity of this layer resulting in immature dentin formation offibrous origin. Increased activity of fibroblasts in the pulp tissue is an indication that this might be ended with fibrosis of the pulp. Further study to the effect of intensified light on the pulp tissue for a more extended period is needed.
References
- Labrie D, Richard J, Price BEM (2011) Evaluation of Ocular Hazards from 4 Types of Curing Lights. J Can Dent Assoc 77: b116.
- Zhi-Chun Zhao, Ying Zhou, Gang Tan, Juan Li (2004) Research progress about preventing blue light on eyes. International J of ophthalmology 11(12): 1999-2003.
- Bruzell ME, Jacobsen N, Pettersen HN (2004) Health hazards associated with curing light in the dental clinic. Clinic Oral Invest 8: 113-117.
- (1985) Council on Dental Materials, Instruments, and Equipment. Visible light-cured composites and activating units. JADA pp. 110.
- Goodis HE White JM, Gamm B, Watanabe L (1990) Pulp chamber temperature changes with visible-light-cured composites in vitro Dental Materials 6(2): 99-102.
- Ruge WAM, Bruder E (2008) Current concepts of enzyme histochemistry in modern pathology. Pathobiology 75(4): 233-243.
- William A (2008) Meier-Ruge Elisabeth Bruder Current Concepts of Enzyme Histochemistry in Modern Pathology. Pathobiology 75(4): 233-243.
- Yoshino F, Yoshida A (2018) Effects of blue-light irradiation during dental treatment. .Japanese Dental Science Review 54(4): 160-168.
- Isabel CCM (2012) post-operative sensitivity after direct resin composite restoration IJRD p. 1.
- Mamalis A, Garcha M, Jared Jagdeo J (2015) Light Emitting Diode-Generated Blue Light Modulates Fibrosis Characteristics: Fibroblast Proliferation, Migration Speed, and Reactive Oxygen Species Generation Lasers Surg Med 47(2): 210-215.
- Ieda N, Hotta Y, Miyata N, Kimura K, Nakagawa H (2014) Photomanipulation of Vasodilation with a Blue-Light-Controllable Nitric Oxide Releaser. Journal of the American Chemical Society 136(19): 7085-7091.
- Yoshida A, Yoshino F, Makita T, Maehata Y, Higashi Y, et al. (2013) Reactive oxygen species production in mitochondria of human gingival fibroblast induced by blue light irradiation. J Photochem 129 Photobiol B Biol 129: 1-5.
- Yoshida A, Ogura YS, Takahashi SW, Takahashi SS, Toyama T, et al. (2015) Blue light irradiation-induced oxidative stress in vivo via R.O.S. generation in rat gingival tissue. Photochem Photobiol B Biol 151: 48-53.
- Martin S, Lin H, Ejimadu C, Lee T (2015) Tissue-nonspecific alkaline phosphatase as a target of sFRP2 in cardiac fibroblasts. Am J Physiol Cell Physiol 309(3).
- Zhu T, Wu Y, Zhou X, Yang Y, Wang Y (2019) Irradiation by blue light-emitting diode enhances osteogenic differentiation in gingival mesenchymal stem cells in vitro. Lasers in Medical Science 34(7): 1473-1481.
- Abe T, Abe Y, Aida, Y Hara Y (2001) Extracellular matrix regulates induction of alkaline phosphatase expression by ascorbic acid in human fibroblasts. Journal of Cellular e Physiology 189(2): 144-151.
- Alonso LRJ, Turrioni SPA, Basso GF, Costa SAC, Hebling J (2016) Synthesis of dental matrix proteins and viability of odontoblast-like cells irradiated with blue LED. Lasers in Medical Science 31: 523-530.
- Perticone F, Perticone M, Maio R, Sciacqua A, Andreucci M, et al. (2015) Alkaline Phosphatase Negatively Affects Endothelium-Dependent Vasodilation in Naïve Hypertensive Patients Hypertension 66(4): 874-880.
- Yoshino F, Yoshida A (2018) Effects of blue-light irradiation during dental treatment. Jpn Dent Sci Rev 54(4): 160-168.
- Anke Suter, Vincent Everts, Alan Boyde, Sheila J Jones, Renate Lüllmann-Rauch, et al. (2001) Overlapping functions of lysosomal acid phosphatase (LAP) and tartrate-resistant acid phosphatase (Acp5) revealed by doubly deficient mice. Development 128: 4899-4910.
- Bull H, Murray PG, Thomas D, Fraser AM, Nelson PN (2002) Acid phosphatases. Mol Pathol 55(2): 65-72.
- Tian P (2016) Research Advances in Succinate Dehydrogenase. Biotechnology: An Indian Journal Review 12(5).
- Rutter J, Winge DR, Schiffman JD (2010) Succinate dehydrogenase – Assembly, regulation, and role in human disease. Mitochondrion 10(4): 393-401.
- Wu J, Seregard S, Algvere P (2006) Photochemical Damage of the Retina. Survey of Ophthalmology 51(5): 461-481.






























