Marcello Ferrari Annual Variations In Vitamin D Levels And Exercise Capacity In Italian Amateur Cyclists
Marcello Ferrari*
Department of Sports Medicine, University of Verona, Italy
Submission: February 10, 2019; Published: February 25, 2019
*Corresponding author: Marcello Ferrari, Department of Sports Medicine, University of Verona, Italy
How to cite this article: Marcello Ferrari. Marcello Ferrari Annual Variations In Vitamin D Levels And Exercise Capacity In Italian Amateur Cyclists. J Phy Fit Treatment & Sports. 2019; 6(1): 555679. DOI:10.19080/JPFMTS.2018.05.555679
Introduction
Vitamin D is a steroid hormone which plays a pivotal role in bone mineral homeostasis and is involved in the regulation of a large number of physiological processes, such as immune function, protein synthesis, inflammatory response and cell growth [1-4]. Indeed, Vitamin D is, together with testosterone, one of most important hormones for function and strength of the muscles, and it could have a significant role in the improvement of performance [5-6]. Deficiency of Vitamin D affects lower limb muscles, that are necessary in simple and frequent actions such as walking and postural balance [7]. In fact, the first symptoms of deficiency of this micronutrient could be muscle weakness, hypotonia and elongation of muscle relaxation time [8], while the relationship between Vitamin D, lung function and physical activity is not completely clear. Recently, several investigations have discovered low levels of Vitamin D in a high percentage of normal subjects and athletes, even in people living in area with extensive sunlight exposition [9]. Furthermore, some studies have shown a relationship between the serum level of 25(OH)D3 hormone and physical performance [10]. As far as the investigations in the young and the athlete are concerned, a European study performed in female adolescents has found that muscle strength is positively associated with circulating concentration of 25-hydroxy-cholecalciferol (25(OH)D) [1].
The same research found that cardiorespiratory fitness and muscle strength were positively associated with 25(OH)D serum concentration in adolescents of both sexes [1]. Furthermore, Koundourakis [11] found a relationship between 25(OH)D and maximal oxygen uptake (VO2 max) in Greek professional football players. On the contrary, a study on football players in Qatar failed to find an association between muscle function and 25(OH)D [2]. Previous studies have examined levels and association between 25(OH)D and performance in different kinds of sports such as: football [7], swimming [3] and hockey on ice [4]. By contrast, there have been very few investigations about Vitamin D values in cyclists, and no study has considered athletes practicing this sport at our latitudes. Our study was performed in a group of male cyclists exercising in an area of Northern Italy and was aimed to measure circulating levels of 25(OH)D, Parathyroid hormone (PTH) at the beginning and the end of the sport season. We intended also to evaluate the possible association between hormone concentrations, physical performance and ventilatory efficiency.
Materials and Methods
Participants
We studied a group of 37 healthy male Caucasian amateur cyclists, living in Veneto, a region of Northern Italy. They were examined at the Unit of Sports Medicine, Department of Internal Medicine, University of Verona.
Design
The study was performed, at the beginning and at the end of the cyclist season. The first part took place in March, at the beginning of spring that in Northern Italy is linked to an increase in heat and duration of the sun during the day, while the second portion was performed in September towards the end of summer. In each experimental period, testing consisted of anthropometric measurements (i.e. height, body weight, body composition), blood testing for the assessment of Vitamin D and PTH levels, and cardiopulmonary test (CPET). During the three days before each experimental session, athletes were informed to avoid caffeine or alcohol beverages. All cyclists were familiarized with the testing protocol, as they had been previously tested with the same procedures on several occasions.
Ethics Statement
Before testing, the aim of the study and the testing procedures were explained in detail to all participants, and written informed consent was obtained. The study was performed in strict accordance with the ethical guidelines of the Helsinki Declaration and was approved by the Ethical Scientific Committee of the Sport Medicine Centre of University of Verona, Italy.
Anthropometrics Measurements and Body Composition
Participants were weighted in short-sleeved tee-shirt, shorts, and socks; stature was measured with a stadiometer to the nearest cm; BMI was calculated as body mass (kg)/stature(m2). For body composition the bioelectrical impedance analysis was utilized, by using an Akern 101 instrument (Pontassieve, Florence, Italy). The device operates at a single frequency (50 kHz) and produces an alternating current of 800 microamperes through the body. The bioelectrical measures considered in this study are the resistance and reactance, from which fat mass (FM; kg) and fat free fass (FFM; kg) were derived using the regression formulas supplied by the manufacturer.
Lung Function Test
All cyclists underwent to lung function test with determination of the forced vital capacity (FVC; l), forced expiratory volume in the first second (FEV1; l / sec) and the ratio of Tiffeneau index (FEV1/FVC). All parameters were determined by using a portable turbine spirometer (Cosmed, Rome, Italy). The values obtained were compared with the theoretical ones using data from Quanjer [12].
Cardiopulmonary Exercise Testing
At the beginning and at the end of the season the cyclists performed an incremental maximal exercise test on a cycle ergometer with electromagnetic brake (Lode Corival). The breath by breath analysis of the expired gases and ventilation was performed by using ZAN 600 (ZAN MESSGERAETE GmbH, Oberthulva, Germany), which allows the instantaneous measurement of O2 and CO2 in the exhaled air. Before each test, the gas analyzer system was calibrated with two different concentrations of gas (15% O2 and 5% CO2, 21% O2 and 0% CO2). In the morning of the test, cyclists were asked to have a light breakfast and to abstain from caffeine and beverages. The protocol of the test was as follows: after a resting phase of 5 minutes, the subject underwent to an incremental ramp protocol, with an initial load of 40 watts and subsequent increments of 40 watts/minute, until exhaustion. During the test 12-lead ECG was recorded. In resting condition and during the test the following variables were recorded: Oxygen uptake (VO2, L/min), CO2 production (VCO2, L/ min), Ventilation (VE, L/min), Maximal heart rate (HR max). The results were expressed as VO2max (measured value in the 10 final seconds at the highest level reached by the patient), VO2 max/kg, and maximal Watt (Watt max, the maximum load reached at the end of the test). Also, the ventilatory equivalents for O2 and CO2 (VE/VO2 and VE/VCO2) were calculated. The VO2max and Watt max were compared with normal values [13]. A test was considered maximal when three of the following criteria were satisfied at the time of: the plateau of VO2 at peak exercise, the RER (VO2/VCO2)> 1.10, the peak heart rate ≥ 85% of the theoretical maximum frequency predicted by age and sex and reached exhaustion.
Vitamin D and PTH Measurements
25(OH)D serum concentration was measured by direct, chemiluminescent, type-competitive immunoassay (CLIA; Liaison, original assay, DiaSorin, Stillwater, MN, USA), which evaluate a range from 7- 150 ng / ml. The sensitivity of the method is 7 ng / ml. The coefficients of variation within and between assay are respectively 10.8% and 13.9%, and the normality range is between 30-80 ng / ml. PTH was measured by two-site immunoradiometric assay (Nichols Institute Diagnostics, San Juan Capistrano, CA); the coefficients of variation of the method are between 5.1% and 8.2%, and the normality range is between 10 -72 pg / ml.
Statistical Analysis
Results are reported as mean ± standard deviation (mean, SD) calculated by conventional procedures unless otherwise stated. The values of the different parameters measured at the beginning and at the end of the season were compared using t-tests for unpaired data. The relationships between serum hormone concentrations on one hand, and anthropometric, body composition, lung function and exercise parameters on the other, were evaluated by the Pearson coefficient. A linear regression model, considering the CPET parameters associated to Vitamin D at the univariate analysis as dependent variables, and age, FFM index, FEV1/FVC, VO2max/kg and Vitamin D values as determinants, was used in order to investigate the possible independent association between hormone serum levels and physical performance parameters. The level of significance was set at p<0.05. All calculations were performed using Microsoft Excel (Microsoft, Redmond, WA, USA) and SPSS software (version 20,0; IBM; SSPS, Chicago, IL, USA).
Results
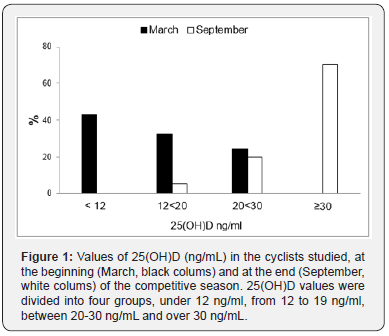
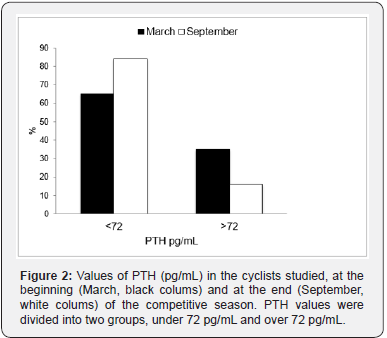
Thirty-seven male cyclists (22-59 years, 1 smoker and 17 former smokers, BMI 24.7 ± 2.3) were studied. The anthropometric parameters did not change significantly between March and September. In lung volumes and the most relevant CPET parameters at the beginning and the end of the season are reported. No variation of FEV1 and VC were found between March and September. On the contrary, a significant increase in VO2 max (p<0.001) and Watt max (p=0.001) were found. Also, VE max, VE/ VO2 max and VE/VCO2 max were significantly higher in September (all p<0.001). As far as Vitamin D serum concentration was concerned, we divided participants into 4 groups, considering ≥30ng/ml normal values (Group 1), and concentrations between 29 and 20 ng/ml (Group 2), 19 and 12 (Group 3) and <12 ng/ ml (Group 4) indicative of insufficiency, deficiency and severe deficiency, respectively [8,9,14]. Figure 1 shows the percentage of subjects in each group in March and September. In March, the average levels of 25 (OH) D were 14.9 ± 6.5 ng/ml, and no athlete showed normal values. Nine athletes (24.3%) were in group 2, 12 (32.4%) in group 3, while the highest number of cyclists 16 (43.3%) were in group 4. At the same time, mean PTH circulating levels were 70.4 ± 26.7 pg/ml, and 13 athletes (35.1%) showed increased PTH concentration, i.e. PTH concentrations above the range normally considered in our laboratory (72 pg/ml) (Figure 2). In September, mean vitamin D serum levels (38.5±10.6 ng/ml) were significantly higher when compared to March (p<0.001). Over 70% of cyclists were in group 1, 7 (18.9%) in group 2, 2 (5.4%) in group 3 and none in group 4. A fairly high percentage of cyclists (16.2%), although lower than that recorded in March, presented PTH serum levels indicative of high concentration.
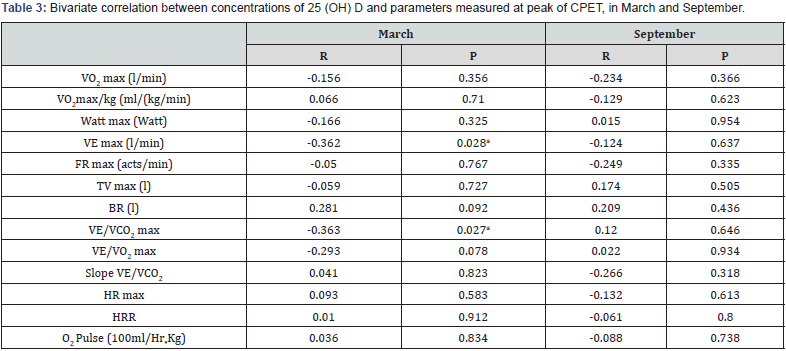
Aiming at verifying whether 25(OH)D levels were associated with anthropometric parameters, we correlated hormone serum concentration with weight, height, BMI, FFM and FM in a bivariate analysis model. In March, a negative relationship was found between vitamin D and FM (r=-0.336, p=0.042) whereas no significant association was found in September. In March, no relationship was found between vitamin D serum levels and VO2 max, Watt max, VE max, FC max and VE/VO2 max. On the contrary, a negative relationship with VE/VCO2 max (r=-0.363, p=0.027) and VE max (r=-0.362; p=0.028) (Figure 3) was found. No significant association between the same parameters was found in September. In a linear regression model, considering VE/VCO2 max measured in March as dependent variable, and age, fat free mass (FFM index), airways patency (FEV1/FVC), physical performance (VO2max/kg) and Vitamin D values as possible determinants, only serum 25(OH)D and VO2max/kg resulted as independent predictors (p=0.028 and p=0.020, respectively) (Tables 1-3).
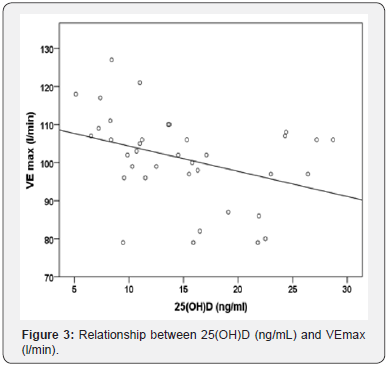
(Maximal ventilation during the cycloergometric test). R=0.362, p=0.028.
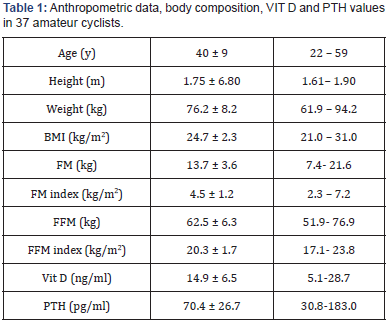
BMI (Body Mass Index); FM (Fat Mass); FM index (Fat Mass/height2); FFM (Fat Free Mass); FFM index; (Fat Free Mass/height2); Vit D (Vitamin D); PTH (Parathyroid hormone).
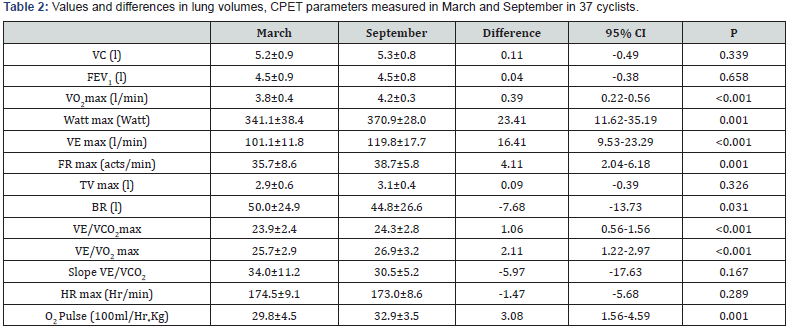
VC (Vital capacity); FEV1 (Expiratory volume.); VO2 max (Maximum oxygen consumption); VE max (Maximal ventilation consumption); FR max (Maximal frequency respiration); TV max (Maximal tidal volume); BR (Respiratory reserve); VE/VCO2 max (CO2 maximal ventilatory equivalent); VE/VO2 max (O2 maximal ventilatory equivalent); HR max (Maximal Heart rate); O2 Pulse (Maximal O2 consumption to heart rate).
Note: A denotes a significant difference correlation with p<0,05.
VO2max (O2 maximum consumption); VE max (Maximal ventilation under effort); FR max (Maximal respiratory frequency); TVmax (Maximal tidal volume); BR (Respiratory reserve); VE/VCO2 max (CO2 maximal ventilatory equivalent); VE/VO2 max (O2 maximal ventilatory equivalent); HR max (Maximal heart rate frequency);
HRR (Heart rate); O2 Pulse (O2 consumption to heart rate).
Discussion
The most important result in this study was the finding of low levels of 25(OH)D in all the athletes at the beginning of the sport season. Moreover, a high percentage of cyclists (43.3%) presented hormone values under 12 ng/ml, indicative of a severe deficit. Serum concentrations of Vitamin D improved significantly through the summer season, even if they remained lower than normal range in a great percentage of athletes. The detection of low levels of Vitamin D has no clear explanation and could be linked to different causes. Firstly, the deficit could be attributed to the place of residence of our athletes. All the subjects live in the Veneto, region of Northern-Eastern Italy. This area is located at 46° latitude. In winter the zenith inclination of the sun becomes more indirect in all regions over 35° latitude, and, consequently, UVB radiation is mostly absorbed or reflected by the atmosphere [15]. It is worth mentioning that the concentrations of airborne dust in this area are among the highest in the world [15]; this could be another reason for filtration of ultraviolet rays. Vitamin D from the diet is an important factor contributing to circulating levels of the hormone [16]. Unfortunately, we have no data on Vitamin D intake in the cyclists studied but we speculate that they would assume low quantities of the hormone, since the typical Mediterranean diet is lacking in it [9]. Moreover, the high ingestion of fibers and phytates, often observed in athletes, could also have reduced the absorption [17]. Another factor that could justify the low hormone concentrations, could be an accelerated catabolism of Vitamin D, related to intense physical exercise. In line with this hypothesis, a recent study demonstrated a decrease in circulating levels of Vitamin D after a period of combat training in military personnel [18].
There is a larger body of research into 25(OH)D deficit in the general population [6-9,19], compared to the studies that have examined this problem in athletes [20-25]. Bannert et al. [22] studying teenager gymnastics, found Vitamin D values lower than normal range in 77% of the sample. A high percentage of hypovitaminosis D was also found in Finnish athletes [23]. Bruce et al. [24] in a study performed on male athletes in the Middle- East, demonstrated that 25(OH)D deficiency is frequent even at those latitudes. In a small study conducted on seven cyclists, a 25(OH)D mean value of 32 ng/ml was found, surprisingly low for a sport performed in the open air [25]. A possible explanation for the negative relationship between circulating levels of 25(OH)D and FM (r=-0.336; p=0.042), is the fact that 25(OH)D, a lipophilic substance, is trapped in adipose tissue [26]. This hypothesis is supported by the finding that obese subjects often present circulating levels of 25(OH)D lower than normal range [27]. Taking into account the low Vitamin D levels, it was expected that a high percentage of our athletes would have secondary increased PTH concentration either in March (35.1%) or in September (16%). In these subjects, an increase in bone turnover is predictable. The increase in PTH could also affect the functioning and quality of muscles, by inducing atrophy of type II fibers by increasing catabolism of proteins, and by reducing intracellular content of phosphates, creatine-phosphate and calcium-ATPase in muscle cells [28]. Recent studies show a reduction in bone mineral density in cyclists, in comparison with sedentary controls or athletes of other sports [29-30]. Moreover, Barry & Kohrt demonstrated a decrease in bone mineral density during one year of training and competitions, in athletes practicing road cycling [31]. These authors suggested that many factors may contribute to bone mineral loss, such as down regulation of sexual hormones, together with an increase in stress hormones and proinflammatory cytokines [31]. We hypothesize that the reduction of bone mass in cyclists could be also due to the low levels of Vitamin D and to secondary high levels of PTH, frequent findings in our athletes.
Previous studies on athletes, mostly performed in north Europe, demonstrated the ergogenic effect of ultraviolet radiation exposure, leading to the speculation that this effect was secondary to increased Vitamin D synthesis. In contrast with this hypothesis, our study doesn’t show any relationship between circulating levels of 25(OH)D and physical performance, expressed as peak VO2 or maximal Watt. Our results also disagree with the results obtained by Koundurakis, who recently reported an association between vitamin D levels [10], muscle strenght and peak VO2 in Greek professional football players. These contrasting results could be attributed to the different types of sport practiced by our subjects and the Greek athletes. In our study the cyclists were not professional and the lower mean values of Vitamin D in our subjects may justify the contrasting finding; the narrow range of 25(OH)D levels may make it difficult to demonstrate a relationship between the hormone and physical performance parameters. An interesting and original finding of our study is the statistically significant negative relationship between 25(OH)D serum levels and VE/VCO2.
Our result could be related to a reduction in the sensitivity of respiratory centers to CO2 or to lactic acid, induced by the Vitamin D. It is also possible that a reflex from muscles could influence ventilation and VE/VCO2 ratio (32) and that Vitamin D could modulate this reflex during exercise through its positive effect on muscle strength and function. The above mentioned hypotheses are merely speculative, and neither our results, nor data from literature allow any definitive conclusions. The fact that no relationship between Vitamin D and VE/VCO2 was found in September, could be due to the confounding effect of training. Our study has some limits. First of all, our athletes presented a narrow range of Vitamin D values, especially in March, a fact that makes it difficult to evaluate correlations with parameters of physical performance. Another limit might be the fact that we didn’t collect a precise dietary information from our athletes, making it impossible to determine exactly whether the low levels of 25(OH) D were subsequent to a reduced oral intake and the increase linked to supplementation. Furthermore, training load or time spent outdoors were not quantified. Finally, the cross-sectional nature of the study doesn’t allow us to establish if a cause-effect relationship between 25(OH)D levels and ventilatory parameters.
Conclusion
Hypovitaminosis D, often associated with an increase in PTH, is a frequent finding in cyclists practicing outdoor activities at our latitude, both in early spring and at the end of training season. This indicates the need for Vitamin D supplementation in this group of athletes. 25(OH)D levels do not correlate with aerobic power parameters, whereas they are inversely related to VE/ VCO2, suggesting a possible modulating effect of vitamin D on ventilation.
References
- Valtueña J, Gracia-MarcoI L, Huybrechts C (2013) Cardio respiratory fitness in males, and upper limbs muscular strength in females, are positively related with 25 hydroxyvitamin D plasma concentrations in European adolescent: the HELENA study. QJM 106 (9): 809-821.
- Maroon JC, Mathyssek CM, Bost JW (2015) Vitamin D profile in National Football League players. Am J Sports Med 43(5): 1241-1245.
- Dubnov-Raz G, Livne N, Raz R, Cohen A (2014) Vitamin D Supplementation and Physical Performance in Adolescent Swimmers. Int J Sport Nutr Exerc Metab 25(4): 317-325.
- Fitzgerald JS, Peterson BJ, Wilson PB (2015) Vitamin D status is associated with adiposity in male ice hockey players. Med Sci Sports Exerc 47(3): 655-661.
- Bartoszewska M, Kamboj M, Patel DR (2010) Vitamin D, muscle function and exercise performance. Pediatr Clin North Am 57(3): 849-861.
- Ooms ME, Lips P, Roos JC (1995) Vitamin D status and sex hormone binding globulin: determinants of bone turnover and bone mineral density in elderly women. J Bone Miner Res 10(8): 1177-1184.
- Hamilton B, Whiteley R, Farooq A (2014) Vitamin D concentration in 342 professional football players and association with lower limb isokinetic function. J Sci Med Sport 17(1): 139-143.
- De Rui M, Toffanello ED, Veronese N (2014) Vitamin D deficiency and leisure time activities in the elderly: are all pastimes the same? Plos One 9(4): e94805.
- Lips P (2010) Worldwide status of vitamin D nutrition J Steroid Biochem Mol Biol 121(1-2): 297-300.
- Afrooz Ardestani, Beth Parker, Shishir Mathur (2011) Relation of Vitamin D Level to Maximal Oxygen Uptake in Adults. Am J Cardiol 107(8): 1246-1249.
- Koundourakis NE, Androulakis NE, Malliaraki N (2014) Vitamin D and exercise performance in professional soccer players. Plos One 9: e101659.
- Quanjer PH (1983) Standardized lung function testing. Report working party. European Community for Coal and Steel. Bull Europ Physiopath Respir 19: 1-82.
- Jones NL, Makrides L, Hitchcock C (2003) Normal standards for an incremental progressive cycle ergometer test. Am Rev Resp Dis 131: 700-708.
- Allison RJ, Close GL, Farooq A (2015) Severely vitamin D-deficient athletes present smaller hearts than sufficient athletes. European J Prev Cardiol 22(4): 535-542.
- Cadum E. Berti G, Biggeri A, Bisanti (2009) The results of EpiAir and the national and international literature. Epidemiol Prev 33( 6 Suppl 1): 113-9; 123-43.
- National Institutes of Health. Vitamin D fact sheet for health professionals.
- Freisling H, Fahey MT, Moskal A (2010) Region-specific nutrient intake patterns exhibit a geographical gradient within and between European Countries. J Nutr 140: 1280-1286.
- Andersen NE, Karl JP, Cable SJ (2010) Vitamin D status in female military personnel during combat training. J Int Soc Sports Nutr 14: 7-38.
- Snijder MB, Van Schoor NM, Pluijm SM (2006) Vitamin D status in relation to one-year risk of recurrent falling in older men and women. J Clin Endocrinol Metab 91: 2980-2985.
- Hamilton B, Whiteley R, Farooq A, Chalabi H (2014) Vitamin D concentration in 342 professional football playersand association with limb isokinetic function. J Sci Med Sport 17: 139-143.
- Bannert N, Starke I, Mohnike K (1991) Parameters of mineral metabolism in children and adolescents in athletic training. Kinderarztl Prax 59(5): 153-156.
- Lehtonen-Veromaa M, Möttönen T, Irjala K (1999) Vitamin D intake is low and hypovitaminosis D common in healthy 9- to 15-year-old Finnish girls. Eur J Clin Nutr 53(9): 746-751.
- Bruce H, Grantham J, Racinais S (2010) Vitamin D deficiency is endemic in Middle Eastern sportsmen. Public Health Nutr 15: 1-7.
- Maïmoun L, Manetta J, Couret I (2006) The intensity level of physical exercise and the bone metabolism response. Int J Sports Med 27(2): 105-111.
- Cannell JJ, Hollis BW (2008) Use of vitamin D in clinical practice. Altern Med Rev 13(1): 6-20.
- Lips P (2007) Vitamin D status and nutrition in Europe and Asia. J Steroid Biochem Mol Biol 103(3-5): 620-625.
- Patten BM, Bilezikian JP, Mallette LE (1974) Neuromuscular disease in primary hyperparathyroidism. Ann Intern Med 80(2): 182-193.
- Nichols JF, Palmer JE, Levy SS (2003) Low bone mineral density in highly trained male master cyclists. Osteoporos Int 14(8): 644-649.
- Nagle KB, Brooks MA (2011) A systematic review of bone health in cyclists. Sports Health 3(3): 235-243.
- Barry DW, Kohrt WM (2008) BMD decreases over the course of a year in competitive male cyclists. J Bone Miner Res 23(4): 484-491.
- Lanteri P, Lombardi G, Colombini A (2013) Vitamin D in exercise: physiologic and analytical concerns. Clin Chim Acta 415: 45-53.






























