Rat Brain Acetyl Cholinesterase as a Biomarker of Cadmium Induced Neurotoxicity
Vivek Kumar Gupta1, Abhishek Kumar1, Nikhat Jamal Siddiqi2 and Bechan Sharma1*
1Department of Biochemistry, University of Allahabad, India
2Department of Biochemistry, King Saud University, Saudi Arabia
Submission: November 10, 2015; Published: January 08, 2016
*Corresponding author: Bechan Sharma, Department of Biochemistry, Faculty of Science, University of Allahabad, Allahabad, 211002, India, Email: bechansharma@gmail.com
How to cite this article: Vivek K G, Abhishek K, Nikhat J S, Bechan S. Rat Brain Acetyl Cholinesterase as a Biomarker of Cadmium Induced Neurotoxicity. Open Access J of Tox. 2015; 1(1): 555553. DOI: 10.19080/OAJT.2016.01.555553
Abstract
Cadmium as potential environmental xenobiotics has been shown to cross blood brain barrier and to adversely influence the activity of AChE and hence the brain functions. In the present study, we have evaluated the impact of cadmium in vitro on the properties of AChE isolated from rat brain. The enzyme was found to be membrane bound and it could be successfully solubilized using 0.2% (v/v) Triton X-100, a nonionic detergent, in the extraction buffer (50mM Phosphate, pH 7.4). The enzyme was found to be highly stable up to one month when stored at -20°C. This enzyme exhibited maximum activity at pH 7.4. AChE when incubated at different temperatures for 5 min, displayed maximum activity at 37°C. Treatment with higher temperatures caused inactivation of enzyme activity. The enzyme followed a simple Michaelis-Menten curve when assayed at varying substrate concentration and yielded Km value to be 0.0370 mM. When a fixed activity of AChE was assayed in presence of different concentrations of cadmium, the enzyme activity was sharply reduced; the IC50 value being about 5.7mM. The enzyme when assayed in presence of cadmium at a concentration equal to its IC50, lost its 50% activity in 77 min (t1/2 ). Cadmium was found to act as a noncompetitive inhibitor to the enzyme. These results suggested that AChE from rat brain may serve as a significant biomarker of cadmium induced neurotoxicity.
Keywords: Acetyl cholinesterase; Brain; Cadmium; Biochemical properties; Inhibition
Abbreviations: RoHS: Restriction on Hazardous Substances; Ach: Acetylcholine; Cd: Cadmium; ATI: Acetylthiocholine Iodide; DTNB: 5, 5'-dithio-bis (2- nitro benzoic acid).
Introduction
Cadmium a most abundant heavy/transition metal, discovered by Fridrich Strohmeyer [1] in 1817 as an impurity in zinc carbonate or colamine. According to the Dmitri Mendeleev's modern periodic table, it falls under group IIB, period 5, having atomic number 48. It is soft bluish white d-block element, chemically similar to the zinc and mercury. This metal does not have any known useful function in the human body and produces harmful effects once it enters the body through inhalation, ingestion and skin contact. Cd can replace iron and copper from a number of cytoplasm and membrane proteins like ferritin, thereby causing rise in the iron and copper ions concentration, which may be associated with the production of oxidative stress via Fenton reaction [2,3]. Cd toxicity may be carried out by the proteins having zinc finger motifs into its structures. Due to the similarity between zinc and cadmium, cadmium can easily replace zinc in biological systems (particularly systems which have -SH containing ligands) and binds ten times more strongly than zinc in biological systems therefore it is difficult to remove. On the other hand, it is also reported that cadmium may also replace magnesium and calcium ions in biological systems, though such replacements are very rare [4,5]. Cadmium is an important component of making batteries, cadmium pigments and coatings and plating and as stabilizers for plastics, chemical stabilizers, metal coatings, alloys, barrier to control neutrons in nuclear fusion, black and white television phosphors, and blue and green phosphors for color television picture tubes, and semiconductors and in molecular biology to block voltage- dependent calcium channels from fluxing calcium ions. Cadmium poisoning is an occupational health hazard associated with industrial processes such as metal plating and the production of nickel-cadmium batteries, pigments, plastics, and other synthetics. Adverse effects of human exposure to cadmium were first established among workers in a cadmium battery factory [6]. Workers are exposed occupationally to cadmium primarily by inhalation of fumes or dust. Some gastrointestinal tract exposure may also occur when dust is removed from the lungs by muco ciliary clearance and subsequently swallowed, or by ingestion of dust on hands, cigarettes, or food [7]. The main sources of exposure to cadmium are specific professional atmospheres, diet, drinking water, and tobacco. The primary route of exposure for the general population is through the diet. Also, many other toxic compounds in cigarette smoke make it difficult to attribute specific adverse effects of smoking to the inhalation of cadmium fumes which can result initially in metal fume fever but may progress to chemical pneumonitis, pulmonary edema, and death. In general, the different forms of cadmium have similar toxicological effects by the inhalation route, although quantitative differences may exist from different absorption and distribution characteristics, particularly for the less soluble cadmium pigments such as cadmium sulfide and cadmium selenium sulfide [8]. Because of its carcinogenic property (classified Number one category of carcinogen by The International Agency for Research on Cancer of USA) cadmium has been banned by the European Union's Restriction on Hazardous Substances (RoHS) which causes cancers of lung, prostrate, pancreas, and kidney. It can also cause osteoporosis, anemia, non hypertrophic emphysema, irreversible renal tubular injury, eosinophilia, anosmia, and chronic rhinitis. The generation of ROS by Cd has been one of the known mechanisms by which this heavy metal induces mutagenesis [9]. Acetyl cholinesterase (AChE, EC 3.1.1.7) or acetyl hydrolase is a serine cholinesterase that hydrolyzes the neurotransmitter acetylcholine to be acetyl Co A. and choline. AChE is found mainly at neuromuscular junctions and cholinergic brain synapses, where its activity serves to terminate synaptic transmission and is synthesized in the endoplasmic reticulum and is then exported towards the cellular surface, where its different molecular/globular forms may be anchored in plasma membrane, attached to the basal lamina (asymmetric collagen-tailed forms) or secreted as soluble molecules (non-globular) forms [10]. It is a key enzyme of nerve impulse transmission and is reported to be inhibited by Cadmium. AChE is an enzyme which occurs at high specific activity in the brain and in nervous tissues and it is readily detected in the membranes of muscles and erythrocytes. The most widely adopted solubilization methods for mammalian brain AChE have involved the application of detergents, particularly Triton X-100, a non-ionic detergent [11]. AChE has been widely exploited as a primary target of action by organophosphorus compounds such as nerve agents [12]. AChE has been the focus of much attention since it was first suggested that it plays an important role in the rapid destruction of acetylcholine (ACh) in a living organ [13]. The catalytic properties, and their occurrence, histochemical localization, and molecular heterogeneity in the different tissues of different animal species have been extensively studied [14-18]. Since cadmium has been found to cross blood brain barrier in mammals and influence the brain functions, it was imperative to evaluate in vitro the impact of cadmium on the biochemical behavior of AChE in order to understand its mechanism of action. In the present study, we have endeavored to characterize AChE from the rat brain and to monitor its interactions cadmium under different experimental conditions. The results have indicated that cadmium may adversely influence brain functions through modulation of AChE activity. Thus, rat brain AChE may be exploited as a key biomarker to assess cadmium toxicity
Materials and Methods
Chemicals
S-acetylthiocholine iodide (ATI) and the coloring reagent 5, 5’-dithio-bis (2- nitro benzoic acid) (DTNB) were procured from Tokyo Chemical Industry Co., Ltd. Tokyo, Japan and SRL Pvt. Ltd. Mumbai, India, respectively. Triton X-100 was purchased from Merck. Bovine serum albumin, Phosphate buffer salts (Sodium dihydrogen orthophosphate and di-Sodium hydrogen phosphate) were obtained from Fisher Scientific and Folin and Ciocalteu's Phenol reagent from Spectrochem Pvt. Ltd. Mumbai, India. All other chemicals were of analytical grade purity.
Animals
Male albino rats of same age group, weighing between 180210 g were selected for all these experiments. Animals obtained from CDRI, Lucknow, India, were housed in propylene cages at temperatures of 30±5°C and 45±5% relative humidity with 12h of light and dark cycle. Animals were fed with standard rat feed available commercially with free access to water. Protocols for care and maintenance of the rats were strictly followed and the study had the approval of institutional ethical committee.
Collection of brain tissues and preparation of homogenates
The healthy rats were sacrificed using mild chloroform anesthesia and cervical dislocation causing minimal pain. The whole brain was quickly excised, washed with isotonic ice cold 0.9% (w/v) NaCl solution, blotted to dryness and weighed. Rat brain tissue homogenate (10% w/v) were made in 50 mM Sodium Phosphate buffer (pH 7.4) containing 0.2% (v/v) Triton X-100 and another without detergent using Potter- Elvehjam homogenizer fitted with a Teflon coated pestle under ice cold condition (4°C). The homogenates were centrifuged at 9000xg for 30 min using REMI refrigerated centrifuge. The supernatants were removed and the pellets were reconstituted in equal volume of homogenizing buffer. Both the supernatants and the pellet's suspensions were used for protein estimation and determination of AChE activity. For determination of IC50, effect of substrate, temperature, pH, and mode of inhibition the supernatant of rat brain homogenate containing Triton X-100 was used.
Protein estimation
The protein was estimated using Folin and Ciocalteu's Phenol reagent [19]. The bovine serum albumin was used as a standard. The absorbance of blue colored complex was monitored at 620nm.
Acetylcholinesterase Assay
The activity of AChE in the brain was determined by method described by Ellman et al. [20]. The reaction mixture (3 ml) in quartz cuvette having 1cm path length contained 0.50mM of ATI, 0.5 mM of DTNB and 50 mM phosphate buffer (pH 7.4). The change in optical density was measured at 412 nm for 3 min at each interval of 30 sec. The AChE activity was calculated using extinction coefficient 13.6x103M-1 cm-1 and expressed as mmoles of acetylthiocholine (ATI) hydrolysed ml-1 min-1 or units (U). The specific activity of enzyme was expressed in U mg-1. The enzyme assays were performed on UV-Visible double beam spectrophotometer (Thermoscientific Spectroscan UV 2700). The catalytic activity is measured by the increase of the yellow anion, 5-thio-2-nitrobenzoate, produced due to reaction of thiocholine with 5, 5’-dithio-bis-(2-nitrobenzoic acid) (DTNB). The assay system without substrate or enzyme was considered as a substrate or enzyme blank, respectively, and any change in absorbance min-1 recorded in this condition were subtracted from the experimental observations.
Effect of substrate concentration on AChE activity
The kinetic parameters Michaelis- Menten constant (km) and maximum velocity (Vmax), were estimated by assaying the enzyme activity using varying substrates concentrations (acetylthiocholine iodide, ATI) (from 0.00 to 2.00 mM) and constant enzyme concentration (132|ig) at room temperature (26±2 °C).
Determination of effect of time on cadmium mediated inhibition of AChE
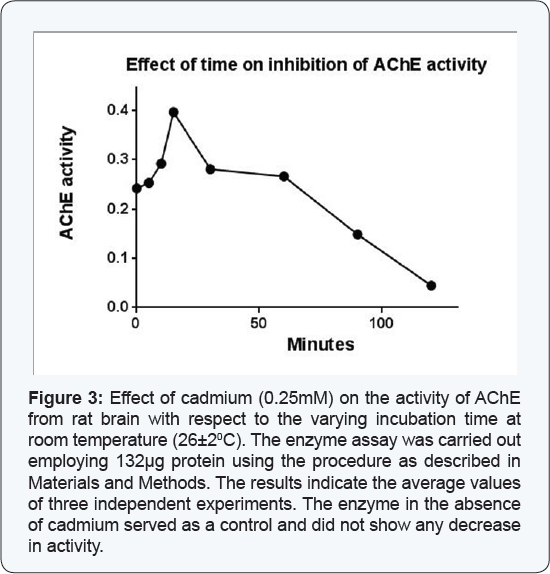
The enzyme (132|ig) was assayed in the presence of 0. 25mM cadmium at various time periods (0 to 120 min) at room temperature (26±20C) and the residual enzyme activity was monitored. The activity of enzyme was also recorded at these time points in absence of Cadmium, which served as a control. The reaction rate measured soon after mixing the enzyme with other reagents without any further incubation was used as zero time reaction. The data of percent residual activity and the time of incubation in min were extrapolated at Y and X-axes, respectively. The t1/2 value (the time at which the enzyme activity remains half of the original under this condition) was calculated from this plot.
Estimation of IC50 value for cadmium
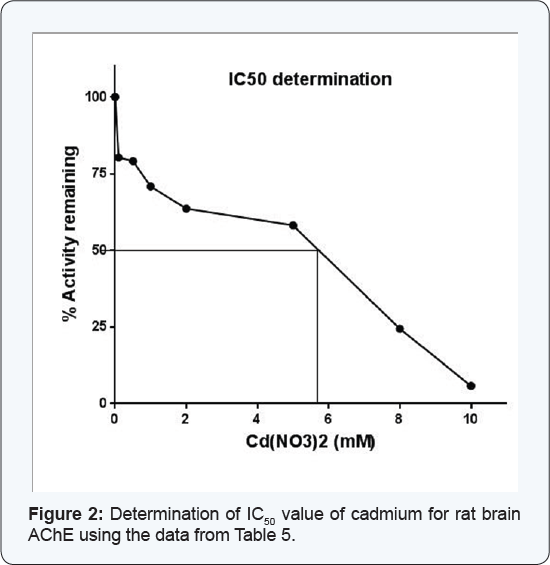
The enzyme (132μg) was assayed in the presence of different concentrations of cadmium nitrate and the residual activity was monitored. The activity recorded in absence of cadmium was considered as 100%. The IC50 value was calculated by extrapolating the data taking percent residual activity on Y-axis and the varying cadmium concentrations at X-axis on a graph.
Determination of mode of inhibition of AChE by cadmium
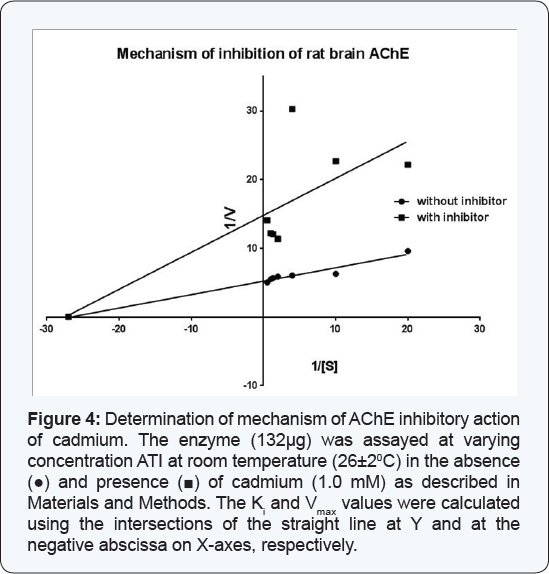
The enzyme (132 μg) was assayed at varying concentrations of ATI at room temperature (26±2°C) in the absence and presence of cadmium (1.0 mM). The Ki and Vmax values were calculated using the intersections by the straight line at Y and at the negative abscissa of X-axes, respectively, of the Lineweaver- Burk's double reciprocal plot. 2.10.
Determination of K value for cadmium in mode of inhibition of AChE from rat brain
The mode of inhibition of enzyme by cadmium was determined by assaying the enzyme mentioned as above using the formula of either
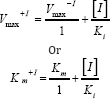
where V max+I and V max-I the maximal velocities of reactions in the presence and absence of cadmium, respectively. Similarly, Km +I and K m denote the Km values in the presence and absence mm of cadmium, respectively. [I] represents the concentration of inhibitor used i.e. 1.0 mM. The Ki value may also be calculated using
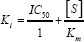
Statistical analysis of data
Statistical analysis of data was performed using Graph Pad Prism version 6 for windows. All values were expressed as mean standard deviation of 3 observations.
Results
Membrane bound nature of AChE from rat brain tissue
The enzyme protein content of brain homogenate from the rat brain tissue were solubilized using a nonionic detergent, 0.2% (v/v) Triton X-100 in phosphate buffer (50mM, pH 7.4). The extent of enzyme activity was more in the detergent solubilized fraction than that of without detergent. These results demonstrated the membrane bound nature of this enzyme. The protein contents in the soluble fractions of these two preparations were also found to be significantly different. The fraction obtained supernatant with Triton X-100 contained 3.30 mg/ml protein as against 1.20 mg/ml in the fraction without treatment with the detergent. The pellet with Triton X-100 contained 1.15 mg/ml protein against 3.15 mg/ml in the fraction without treatment with the detergent, thereby showing solubilization and release of proteins from the pellets in presence of the detergent (Table 1).
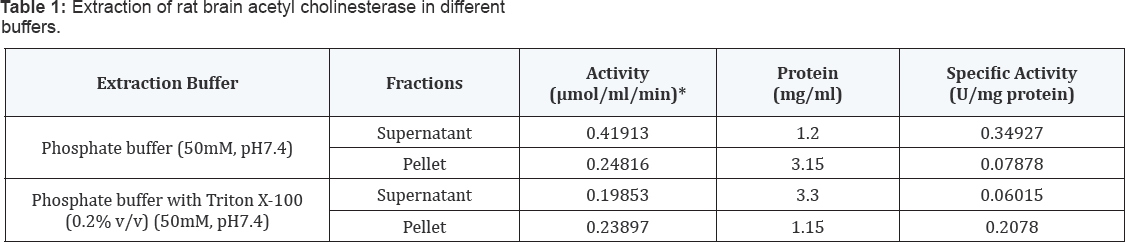
*One unit of the activity of AChE has been defined as the micromoles of substrate hydrolyzed per min per ml.
Stability of enzyme activity after storage at -200C
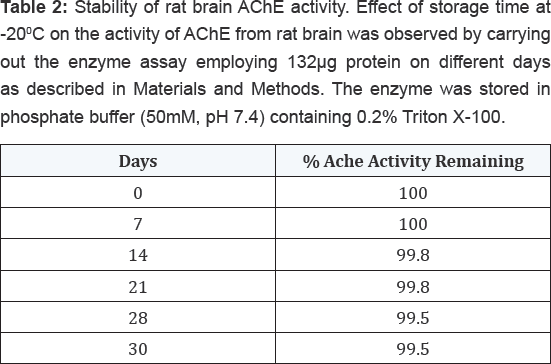
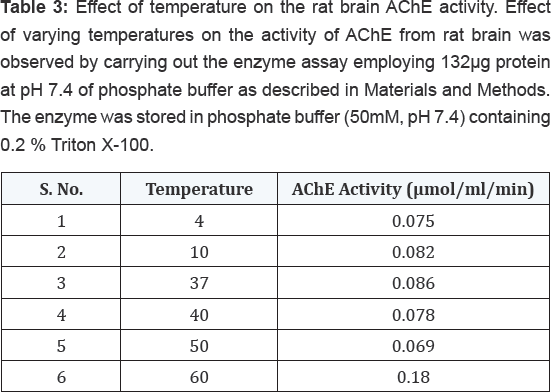
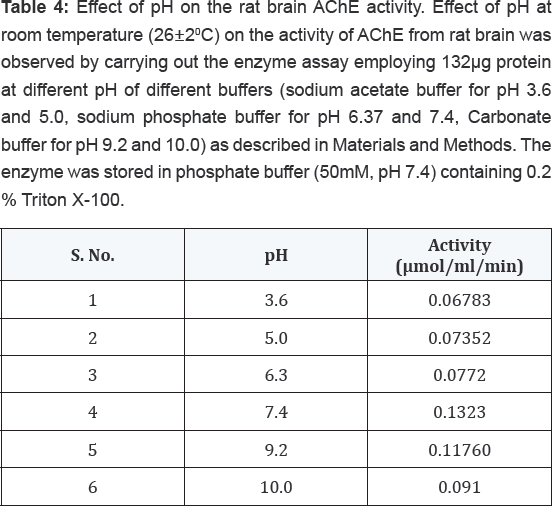
The effect of storage time at -200C on the activity of AChE from rat brain tissue was determined by carrying out the enzyme assay employing 132μg proteins on different days as described in Materials and Methods. The enzyme was stored in phosphate buffer (50mM, pH 7.4) containing 0.2% (v/v) Triton X-100. The results shown in Table 2 indicated that the enzyme was highly stable up to 30 days with no much loss in activity. However, when this enzyme was assayed at varying temperatures, it exhibited maximum activity at 370C followed by gradual loss in its activity after increasing temperature as shown in Table 3. The enzyme was found to be optimally active at pH 7.4 when assayed using buffers of different pH systems under standard assay conditions as shown in Table 4. The buffers of higher pH values displayed inhibitory effect on enzyme activity.
Effect of substrate concentration on the activity of AChE from rat brain tissue
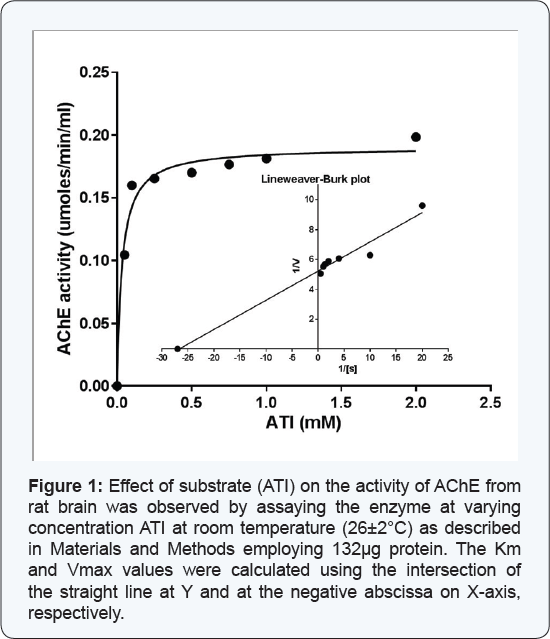
The enzyme (132μg) was assayed at varying concentrations of the substrate (ATI), at room temperature (26±20C). The enzyme activity at corresponding substrate concentration displayed a direct correlation and the results showed a hyperbolic curve (Figure 1). The Lineweaver Burk's double reciprocal plot of the data as shown in Figure 1 demonstrated a straight line which intersects at Y and negative abscissa of X-axes, from where the Vmax and Km values could be calculated; the values being 0.192μmoles ml-1min-1 and 0.037mM, respectively.
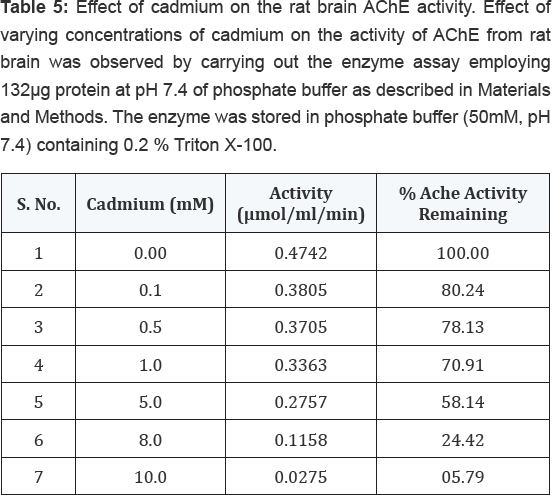
Effect of cadmium on the activity of AChE from rat brain tissue
The enzyme when assayed in presence of varying concentrations of cadmium (0.05-10mM) displayed consistent decrease in its activity (Table 5). When this data was extrapolated using percent residual activity and the cadmium concentrations on Y and X-axes, respectively, the IC50 value of this heavy metal for rat brain AChE could be determined, the value being 5.70mM (Figure 2).
Effect of time on cadmium mediated inhibition of AChE activity from rat brain tissue
The effect of time on the cadmium mediated inhibition of the AChE activity from rat brain at room temperature (26±20C) was monitored by assaying the enzyme (132μg) at different time intervals in absence and presence of the cadmium (0.25mM). The enzyme activity in the absence of cadmium served as a control. The results as presented in Figure 3 demonstrated that the enzyme activity increased up to 15 min and after that it decreased consistently with respect to the increasing incubation time in presence of cadmium. The results from present study also demonstrated that cadmium at a concentration of 0.25mM did not shown any effect on its activity up to 15 min but it shows sharp inhibition of the enzyme in a time dependent manner at 26±20C temperature. The enzyme activity remained about 50% after 77 min of incubation, i.e. the t1/2 time. However, the enzyme did not show any decrease in activity in the absence of cadmium under similar experimental conditions.
Evaluation of mode of inhibition of AChE from rat brain by cadmium
The above experiments indicated that treatment of rat brain AChE with cadmium resulted into decline in its activity. In order to ascertain the mechanism of inhibition of AChE activity by cadmium, the enzyme (132μg) was assayed at varying substrate concentrations in absence and presence of cadmium (1.0mM). The data were used to extrapolate Lineweaver Burk's double reciprocal plot, which developed two straight lines originated from same point on X-axis and intersecting at different points on Y axis (non-competitive inhibition). The V max+I (Vmax value in presence of cadmium) and Ki values were calculated using these points and found to be 0.069 μmoles/ml/min and 0.548mM, respectively. The results are shown in Figure 4.
Discussion
Before an enzyme can be characterized, it must be extracted and solubilized from the selected tissue. Some of the enzymes are membrane-bound and their solubilization has been shown to be achieved by using organic solvents, detergents, proteolytic and lipolytic enzymes [21]. It has been shown that EDTA and tetracaine increased the amount of enzyme extracted from calf brain, and a preliminary studies showed that these compounds had a similar effect on the extraction of enzyme from mouse brain. The results from the present study demonstrated the membrane bound form of rat brain AChE which could be easily solubilized using Triton X- 100. In some other living systems also the membrane bound nature of AChE has been indicated [22-24]. However, AChE has been shown to exist only in a membrane bound form in the human erythrocytes and brain, electric eel, electric fish and certain parasite helminthes [22,23]. The enzyme from these systems has been solubilized by applying both the ionic as well as non-ionic detergents in the phosphate buffer [22]. The Km value for any enzyme is a measure of its affinity towards its substrate. In the present study, the Km value for rat brain AChE was 37μM which was about three times lower than that reported for the enzyme from human brain (107μM), and fetal bovine serum (120 μM) [25]. The rat brain AChE displayed Km about 6times lower than that reported for the analogous enzyme from the human erythrocytes (225μM). The relatively lower Km value for rat brain AChE indicated its enhanced affinity to the substrate as compared to other mammalian systems [22]. The entry of cadmium in mammalian brain has been demonstrated. It is therefore presumed that it may alter brain function. The results from the present study displayed that cadmium caused strong inhibition of rat brain AChE (IC50=5.7mM). These results indicate that cadmium may act as an inducer of toxic stress on the neurotransmission system of rat. In the present study, cadmium inhibited the activity of rat brain AChE in noncompetitive manner when tested in vitro. These results suggest the binding of cadmium at a different site on the enzyme surface other than the active site. No such reports are available from other workers to be used for the sake of the comparison.
Conclusion
The results from the present study indicated the presence of membrane bound form of AChE in the rat brain which could be solubilized employing a non-ionic detergent, Triton X-100. Cadmium sharply inhibited this enzyme at low concentration indicating thereby its strong neurotoxic potential to the mammals. Though the exact mechanism of action of cadmium on brain AChE is not known but this study presented evidence that this heavy metal may inhibit the enzyme in a noncompetitive manner. Thus, the rat brain AChE may serve as a potential biomarker of neurotoxicity induced by cadmium. The information obtained from this study may be useful in proper risk management of cadmium toxicity particularly in those who are occupationally engaged in cadmium infested environment.
References
- Hermann CS (1818) Another letter about the new metal. Annals of Physics 59(2): 113-116.
- Casalino E, Sblano C, Landriscina C (1997) Enzyme activity alteration by cadmium administration to rats: the possibility of iron involvement in lipid peroxidation. Arch Biochem Biophys 346(2): 171-179.
- Valko M, Morris H, Cronin MT (2005) Metals, toxicity and oxidative stress. Curr Med Chem 12(10): 1161-1208.
- Lane TW, Morel FMM (2000) A biological function for cadmium in marine diatoms. Proceedings of the National Academy of Sciences of the United States of America 97(9): 4627-4631.
- Lane TW, Saito MA, George GN, Pickering IJ, Prince RC, et al. (2005) Biochemistry: a cadmium enzyme from a marine diatom. Nature 435(7038): 42.
- Friberg L (1950) Injuries following continued administration of cadmium; preliminary report of a clinical and experimental study Arch Indust Hyg $ Occupational Med 1(4): 458-466.
- Adamsson E, Piscator M, Nogawa K (1979) Pulmonary and gastrointestinal exposure to cadmium oxide dust in a battery factory. Environ Health Perspect 28: 219-222.
- Klimisch HJ (1993) Lung deposition, lung clearance and renal accumulation of inhaled cadmium chloride and cadmium sulphide in rats. Toxicology 12(84): 103-124.
- Filipic M, Fatur T, Vudrag M (2006) Molecular mechanisms of cadmium induced mutagenicity. Hum Exp Toxicol 25(2): 67-77.
- Chatel JM, Vallette FM, Massoulié J, Grassi J (1993) A conformation- dependent monoclonal antibody against active chicken acetylcholinesterase. FEBS Lett 319(1-2): 12-15.
- Rieger F, Vigny M (1976) Solubilisation and physicochemical characterization of rat brain acetylcholinesterase: development and maturation of its molecular forms. J Neurochem 27(1): 121-129.
- Pohanka M (2011) Cholinesterases, a target of pharmacology and toxicology. Biomed Pap Med Fac Univ Palacky Olomouc Czech Repub 155(3): 219-229.
- Dale HH (1914) The action of certain esters of cholme and then relation to muscarme. J Pharmacol Exp Ther 6: 147-190.
- Silver A (1974) The Biology of Cholinesferases North Holland, Amsterdam,”.
- Rosenberry TL (1975) Acetylcholinesterase. Adv Enzymol 43: 103218.
- Trevor AJ, Gordonand MA, Parker KK (1978) Acetylcholinesterase. Life SCI 23: 1209-1220.
- Massoulié J, Bon S (1982) The molecular forms of cholinesterase and acetylcholinesterase in vertebrates. Ann Rev Neurosci 5: 57-106.
- Brimijoin S (1983) Molecular forms of acetylcholmesterase in brain, nerve and muscle Nature, localization and dynamics. Prog Neurobiol 21(4): 291-322.
- Lowry OH, Rosebrough NJ, Farr AL, Randall RJ (1951) Protein measurement with the Folin phenol reagent. J Biol Chem 193(1): 265-275.
- Ellman GL, Courtney KD, Andreas V, Featherstone RM (1961) A new and rapid colorimetric determination of acetylcholinesterase activity. Biochem Pharmacol 7(2): 88-95.
- Hollunger EG, Niklasson BH (1973) The release and molecular state of mammalian brain acetylcholinesterase. J Neurochem 20(3): 821836.
- Ott P (1985) Membranes acetyl cholinesterases: purification, molecular properties and interactions with amphiphilic environments. Biochim. Biophys. Acta 822 (3-4): 375-392.
- Sharma B (1988) Purification and characterization of phosphor fructo kinase and acetyl cholinesterase from Setaria cervi, a filarial parasite. Thesis, Banaras Hindu University, Varanasi.
- Sharma B (1991) In vitro effect of anthelmintics on the motility as well as activity of acetyl-cholinesterase from Setaria cervi, a filarial parasite. Biochem Archives 7: 161-168.
- Ralston JS, Rush RS, Doctor BP, Wolfe AD (1985) Acetylcholinesterase from fetal bovine serum. Purification and characterization of soluble G4 enzyme. J Biol Chem 260(7): 4312-4318.






























